阿尔茨海默氏病(AD),也简称为阿尔茨海默氏症,是一种慢性神经退行性疾病,通常随着时间的推移开始缓慢并逐渐恶化。[1] [2]这是60-70%的痴呆病例的原因。[1] [2]最常见的早期症状是难以记住最近发生的事件。[1]随着疾病的进展,症状可能包括语言问题,迷失方向(包括容易迷路),情绪波动,失去动力,不管理自我护理和行为问题。[1] [2]随着一个人的病情下降,他们经常退出家庭和社会。[1]渐渐地,身体功能丧失,最终导致死亡。[10]虽然进展速度可能不同,但诊断后的典型预期寿命为3至9年。[7] [11]
人们对阿尔茨海默病的病因了解甚少。[1]大约70%的风险被认为是遗传在一个人的父母身上,通常涉及许多基因。[4]其他风险因素包括头部受伤,抑郁和高血压病史。[1]疾病过程与大脑中的斑块和神经原纤维缠结有关[4]。可能的诊断基于疾病史和医学成像和血液检查的认知测试,以排除其他可能的原因。[5]最初的症状经常被误认为正常衰老。[1]需要对脑组织进行检查以确定诊断。[4]精神和体育锻炼,避免肥胖可以降低AD的风险;但是,支持这些建议的证据很薄弱。[4] [12]没有药物或补品可以降低风险。[13]
没有治疗可以阻止或逆转其进展,但有些可能暂时改善症状。[2]受影响的人越来越依赖他人寻求帮助,往往给照顾者带来负担。[14]压力可能包括社会,心理,物质和经济因素。[14]锻炼计划可能对日常生活活动有益,并可能改善结果。[15]痴呆症引起的行为问题或精神病常常用抗精神病药物治疗,但通常不建议这样做,因为早期死亡风险增加几乎没有益处。[16] [17]
2015年,全球约有2980万人患有AD。[2] [8]它通常始于65岁以上的人群,尽管4-5%的病例是早发性阿尔茨海默氏症。[3]它影响了大约6%的65岁及以上的人。[1] 2015年,痴呆症导致约190万人死亡。[9]它最初由德国精神病学家和病理学家Alois Alzheimer在1906年描述,后来以其命名。[18]在发达国家,AD是经济上最昂贵的疾病之一。[19] [20]
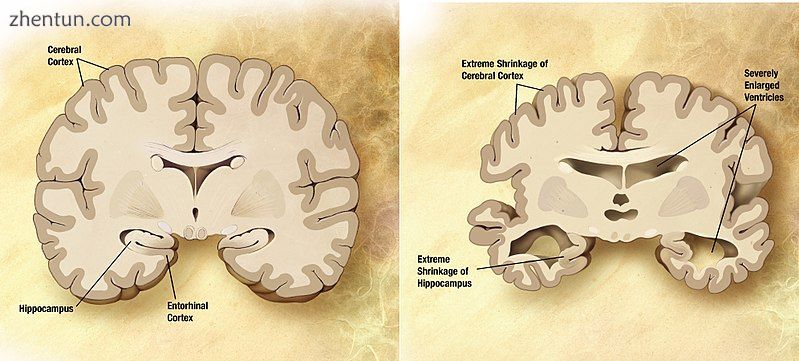
比较正常老年人的大脑(左)和阿尔茨海默氏症患者的大脑(右)。 指出了将两者分开的特征。
目录
1 症状和体征
1.1 痴呆前期
1.2 早期
1.3 中期
1.4 晚期
2 原因
2.1 遗传
2.2 胆碱能假说
2.3 淀粉样蛋白假说
2.4 Tau假设
2.5 其他假设
3 病理生理学
3.1 神经病理学
3.2 生物化学
3.3 疾病机制
4 诊断
4.1 标准
4.2 技术
5 预防
5.1 药物治疗
5.2 生活
5.3 饮食
6 管理
6.1 药物治疗
6.2 心理社会干预
6.3 看护
7 预后
8 流行病学
9 历史
10 社会与文化
10.1 社会成本
10.2 护理负担
10.3 媒体
11 研究方向
11.1 用药
11.2 行为预防
11.3 可能的传播
11.4 感染
11.5 成像
11.6 诊断
12 参考
体征和症状
阿尔茨海默病的阶段[21]
衰老对记忆的影响,但不影响AD
偶尔忘记事情
有时错放物品
轻微的短期记忆丧失
不记得确切的细节
早期阿尔茨海默氏症
不记得健忘的情节
忘记家人或朋友的名字
只有亲密的朋友或亲戚才能注意到变化
在熟悉的情况之外的一些混乱
中期阿尔茨海默氏症
难以记住最近学到的信息
在许多情况下深化混乱
睡眠问题
无法确定其位置
晚期阿尔茨海默氏症
思考能力差
说话有问题
重复相同的对话
更多的虐待、焦虑或偏执
疾病过程分为四个阶段,具有认知和功能障碍的进行模式。
痴呆前期
最初的症状通常被错误地归因于衰老或压力。[22] 详细的神经心理学测试可以揭示在一个人达到诊断AD的临床标准之前长达八年的轻度认知困难。[23] 这些早期症状可能影响日常生活中最复杂的活动。[24] 最显着的缺陷是短期记忆丧失,这表现为难以记住最近学到的事实和无法获得新信息。[23] [25]
注意力,计划,灵活性和抽象思维的执行功能或语义记忆中的损伤(意义记忆和概念关系)的微妙问题也可能是AD早期阶段的症状。[23]在这个阶段可以观察到冷漠,并且在整个疾病过程中仍然是最持久的神经精神症状。[26]抑郁症状,易怒和对细微记忆困难的认识降低也很常见。[27]该疾病的临床前阶段也被称为轻度认知障碍(MCI)。[25]这通常被发现是正常衰老和痴呆之间的过渡阶段。 MCI可出现各种症状,当记忆丧失是主要症状时,它被称为“遗忘性MCI”,并且经常被视为阿尔茨海默病的前驱期。[28]
早期
在患有AD的人中,学习和记忆的不断增加的损害最终导致明确的诊断。在一小部分中,语言,执行功能,感知(失认症)或动作执行(失用症)的困难比记忆问题更为突出。[29] AD不会同等地影响所有内存容量。关于人的生活(情节记忆),事实学习(语义记忆)和内隐记忆(身体对如何做事的记忆,例如使用叉子吃或如何从玻璃杯中饮用)的旧记忆受到影响程度低于新事实或记忆。[30] [31]
语言问题的主要特征是词汇量减少,词语流利性下降,导致口头和书面语言普遍贫困。[29] [32]在这个阶段,患有阿尔茨海默氏症的人通常能够充分地传达基本思想。[29] [32] [33]在执行诸如书写,绘画或穿衣等精细运动任务时,可能会出现某些运动协调和计划困难(失用症),但它们通常不被注意。[29]随着疾病的进展,患有AD的人通常可以独立地继续执行许多任务,但可能需要帮助或监督最具认知要求的活动。[29]
中期
逐步恶化最终会阻碍独立,受试者无法进行日常生活中最常见的活动。[29]由于无法回忆词汇,言语困难变得明显,这导致频繁的不正确的单词替换(paraphasias)。阅读和写作技巧也逐渐丧失。[29] [33]随着时间的推移和AD的进展,复杂的运动序列变得不那么协调,因此下降的风险增加。[29]在这个阶段,记忆问题恶化,这个人可能无法认出近亲。[29]以前完好无损的长期记忆会受损。[29]
行为和神经精神病学变化变得更加普遍。常见的表现是徘徊,烦躁和不稳定的影响,导致哭泣,爆发无预谋的侵犯或抵抗照顾。[29]日落也可以出现。[34]大约30%的AD患者会出现错觉辨别和其他妄想症状。[29]受试者也不了解他们的疾病过程和局限性(anosognosia)。[29]尿失禁可以发展。[29]这些症状会给亲属和照顾者带来压力,这可以通过将人从家庭护理转移到其他长期护理机构来减少。[29] [35]
晚期
在最后阶段,患者完全依赖照顾者。[29]语言被简化为简单的短语甚至单个单词,最终导致语言完全丧失。[29] [33]尽管失去了语言能力,但人们通常能够理解并回归情绪信号。虽然侵略性仍然可以存在,但极度冷漠和疲惫是更常见的症状。患有阿尔茨海默病的人最终将无法独立完成最简单的任务;肌肉质量和活动能力恶化到他们卧床不起、无法自食其力的程度。死亡的原因通常是一个外部因素,如压疮或肺炎的感染,而不是疾病本身。[29]
原因
除了已发现遗传差异的1%至5%的病例外,大多数阿尔茨海默病病例的病因仍然大多未知[36] [37]。试图解释这种疾病的原因存在几种相互竞争的假设。
遗传
根据双胞胎和家庭研究的综述,阿尔茨海默病(及其记忆成分)的遗传遗传率在49%至79%之间[38] [39]。大约0.1%的病例是家族性的常染色体(非性连锁)显性遗传,在65岁之前就已发病。[40]这种疾病形式被称为早发型家族性阿尔茨海默病。大多数常染色体显性家族性AD可归因于三种基因之一的突变:编码淀粉样蛋白前体蛋白(APP)和早老素1和2的基因。[41] APP和早老素基因中的大多数突变增加了一种名为Aβ42的小蛋白的产生,这是老年斑的主要成分。[42]一些突变仅改变Aβ42与其他主要形式之间的比例 - 特别是Aβ40-而不增加Aβ42水平。[43] [44]与常染色体显性遗传性阿尔茨海默病相关的另外两个基因是ABCA7和SORL1。[45]
大多数阿尔茨海默病病例没有表现出常染色体显性遗传,被称为散发性AD,其中环境和遗传差异可能成为危险因素。最著名的遗传风险因素是载脂蛋白E(APOE)的ε4等位基因的遗传。[46] [47] 40%至80%的AD患者至少有一个APOEε4等位基因。[47] APOEε4等位基因在杂合子中将该疾病的风险增加三倍,在纯合子中增加15倍。[40]像许多人类疾病一样,环境影响和遗传修饰因素导致不完全的外显率。例如,某些尼日利亚人群没有显示APOEε4的剂量与其他人群中出现的阿尔茨海默病的发病率或发病年龄之间的关系。[48] [49]早期尝试筛选多达400个与迟发性散发性AD(LOAD)相关的候选基因导致产量低[40] [41]。最近的全基因组关联研究(GWAS)已经发现了19个似乎影响风险的基因区域。[50]这些基因包括:CASS4,CELF1,FERMT2,HLA-DRB5,INPP5D,MEF2C,NME8,PTK2B,SORL1,ZCWPW1,SlC24A4,CLU,PICALM,CR1,BIN1,MS4A,ABCA7,EPHA1和CD2AP。[50]
TREM2基因中的等位基因与患阿尔茨海默病的风险相关,高出3到5倍。[51] [52]建议的作用机制是在TREM2的一些变体中,脑中的白细胞不再能够控制存在的β淀粉样蛋白的量。许多SNP与阿尔茨海默氏症有关,2018年的研究通过将AD分为6类,包括记忆,语言,视觉空间和执行功能,增加了30个SNP。[53]
胆碱能假说
目前最常用的药物疗法所依据的最老的是胆碱能假说[54],其提出AD是由神经递质乙酰胆碱的合成减少引起的。胆碱能假说尚未得到广泛支持,主要是因为用于治疗乙酰胆碱缺乏症的药物治疗并不十分有效。[55]
淀粉样蛋白假说
1991年,淀粉样蛋白假说假定细胞外淀粉样蛋白β(Aβ)沉积是该疾病的根本原因。[56] [57]对这一假设的支持来自21号染色体上淀粉样蛋白前体蛋白(APP)基因的位置,以及具有额外基因拷贝的21三体综合征患者几乎普遍表现出至少最早的症状AD的年龄为40岁。[58] [59]此外,载脂蛋白的特定同种型APOE4是AD的主要遗传风险因子。虽然载脂蛋白可以增强β淀粉样蛋白的分解,但是某些同种型在这项任务中并不是非常有效(例如APOE4),导致大脑中过量的淀粉样蛋白积聚[60]。进一步的证据来自于发现表达人APP基因的突变形式的转基因小鼠发展出纤维状淀粉样斑块和具有空间学习缺陷的阿尔茨海默氏样脑病理学[61]。
发现一种实验性疫苗可以在早期人体试验中清除淀粉样斑块,但它对痴呆没有任何显著影响。[62]研究人员已经将可疑的非斑块Aβ寡聚体(许多单体的聚集体)作为Aβ的主要致病形式。这些有毒低聚物,也称为淀粉样蛋白衍生的可扩散配体(ADDLs),与神经元上的表面受体结合,改变突触的结构,从而破坏神经元的通讯。[63] Aβ寡聚体的一种受体可能是朊病毒蛋白,与疯牛病和相关人类病症Creutzfeldt-Jakob病相关的相同蛋白,因此可能将这些神经退行性疾病的潜在机制与阿尔茨海默病的潜在联系起来[ 64]
在2009年,该理论得到了更新,表明β-淀粉样蛋白的近亲,而不一定是β-淀粉样蛋白本身,可能是该疾病的主要罪魁祸首。该理论认为,淀粉样蛋白相关的机制可以在早年生命的快速生长阶段修复大脑中的神经元连接,这可能是由于晚年与衰老相关的过程引起的,导致阿尔茨海默病的神经元萎缩。[65] N-APP是来自肽N-末端的APP片段,与β-淀粉样蛋白相邻,并且通过相同的酶之一从APP切割。 N-APP通过与称为死亡受体6(DR6,也称为TNFRSF21)的神经元受体结合而触发自毁途径。[65] DR6在受阿尔茨海默氏症影响最严重的人类大脑区域中高度表达,因此N-APP / DR6通路可能在老化的大脑中被劫持而导致损伤。在该模型中,β-淀粉样蛋白通过抑制突触功能起到补充作用。
2017年初,由于一个独立的小组发现“几乎没有找到积极临床效果的机会”,因此停止了verubecestat试验,该试验抑制负责产生β-淀粉样蛋白的β-分泌酶蛋白。[66]
Tau假设
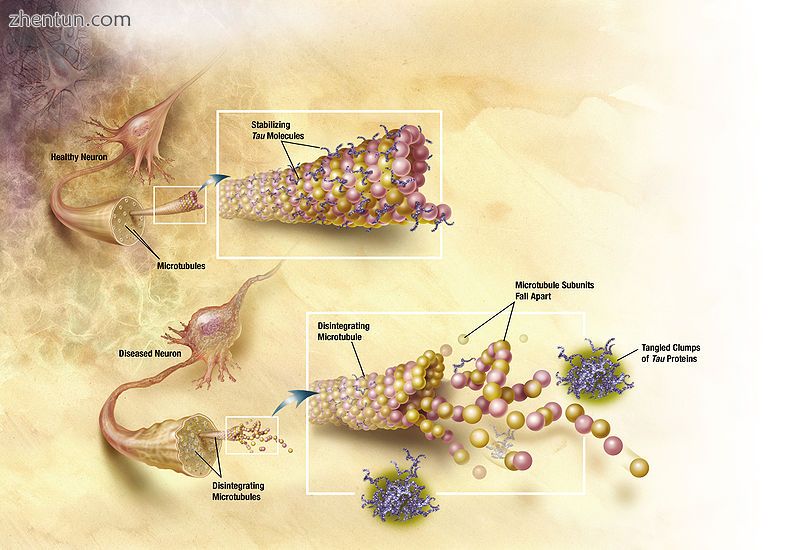
在阿尔茨海默病中,tau蛋白的变化导致脑细胞中微管的分解。
tau假说提出tau蛋白异常引发疾病级联。[57]在该模型中,过度磷酸化的tau开始与tau的其他线程配对。最终,它们在神经细胞体内形成神经原纤维缠结。[67]当这种情况发生时,微管就会崩解,破坏细胞骨架的结构,从而使神经元的运输系统崩溃。[68]这可能首先导致神经元之间的生化通讯失败,然后导致细胞死亡。[69]
其他假设
已经提出了一种神经血管假说,其中指出可能涉及血脑屏障的功能不良。[70]
生物金属(如离子铜,铁和锌)的细胞稳态在AD中被破坏,但仍不清楚这是由蛋白质产生还是引起蛋白质的变化。这些离子影响并受到tau,APP和APOE的影响[71],它们的失调可能引起可能导致病理的氧化应激。[72] [73] [74] [75] [76]其中一些研究的质量受到了批评,[77] [78]并且这种联系仍然存在争议。[79]大多数研究人员不支持与铝的因果联系。[78]
吸烟是AD的重要风险因素。[80]先天免疫系统的系统标志物是晚发性AD的危险因素。[81]
有初步证据表明,接触空气污染可能是导致阿尔茨海默病的一个因素。[82]
牙龈疾病:牙龈疾病中的螺旋体(一种细菌)感染可能引起痴呆,并可能参与阿尔茨海默病的发病机制。[83]真菌感染也可能是一个因素。[84]
一种假设认为,衰老过程中少突胶质细胞及其相关髓鞘的功能障碍会导致轴突损伤,从而导致淀粉样蛋白产生和tau蛋白过度磷酸化作为副作用。[85] [86]
逆向发生是由Barry Reisberg在20世纪80年代提出的关于阿尔茨海默病发展和进展的医学假设。[87]假设是,正如胎儿经历从神经系统开始并以髓鞘形成结束的神经发育过程一样,AD患者的大脑经历了一个逆转神经变性过程,从脱髓鞘和轴突死亡(白质)开始,到死亡结束灰质。[​​88]同样,假设是,当婴儿经历认知发展状态时,AD患者会经历进行性认知障碍的逆向过程。[87] Reisberg开发了一种称为“快速”(功能评估分期工具)的护理评估工具,他说这可以让那些照顾AD患者的人识别疾病进展的阶段,并提供有关每个阶段所需护理类型的建议。[87 ] [89]
与乳糜泻的关系尚不清楚,2019年的一项研究发现CD患者的痴呆总体没有增加,而2018年的一项研究发现与包括AD在内的几种类型的痴呆症有关[90] [91]。
病理生理学
主要文章:阿尔茨海默病的生物化学
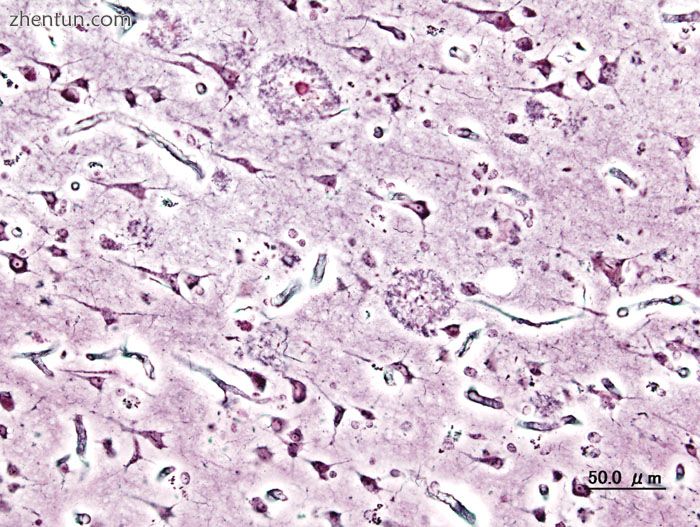
在早老性阿尔茨海默病发病的人的大脑皮质中看到的老年斑的组织病理学图像。银浸渍。
神经病理学
阿尔茨海默病的特征在于大脑皮质和某些皮层下区域的神经元和突触的丧失。这种损失导致受影响区域的严重萎缩,包括颞叶和顶叶的退化,以及额叶皮层和扣带回的部分。[92]变性也存在于脑干核如蓝斑中[93]。使用MRI和PET的研究表明,随着AD从轻度认知障碍进展到阿尔茨海默病,以及与健康老年人的类似图像相比,AD患者特定大脑区域的大小减少。[94] [95]
淀粉样蛋白斑和神经原纤维缠结通过显微镜检查明显可见AD患者的大脑。[96]斑块是致密的,大部分不溶的β-淀粉样蛋白肽沉积物和神经元外部和周围的细胞物质。 Tangles(神经原纤维缠结)是微管相关蛋白tau的聚集体,其已经变得过度磷酸化并在细胞内积累。尽管许多老年人因衰老而出现斑块和缠结,但AD患者的大脑在特定大脑区域(例如颞叶)中的大脑数量更多。[97]路易体在AD患者的大脑中并不罕见。[98]
生物化学
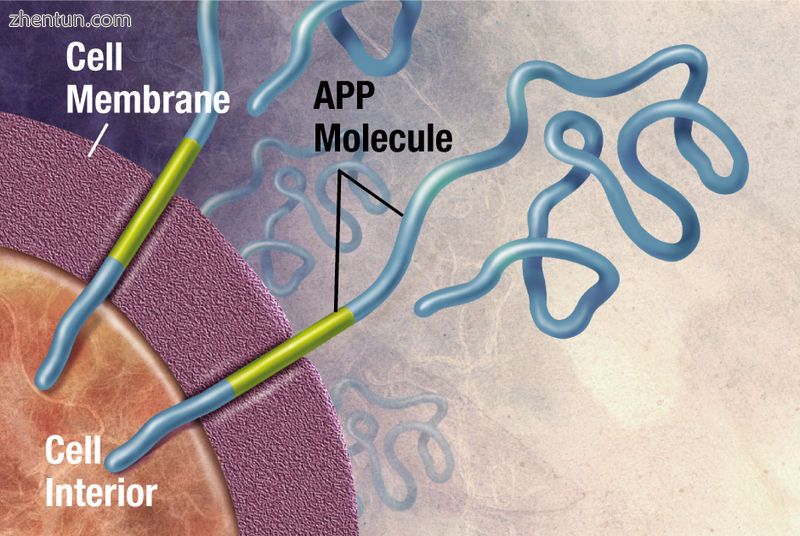
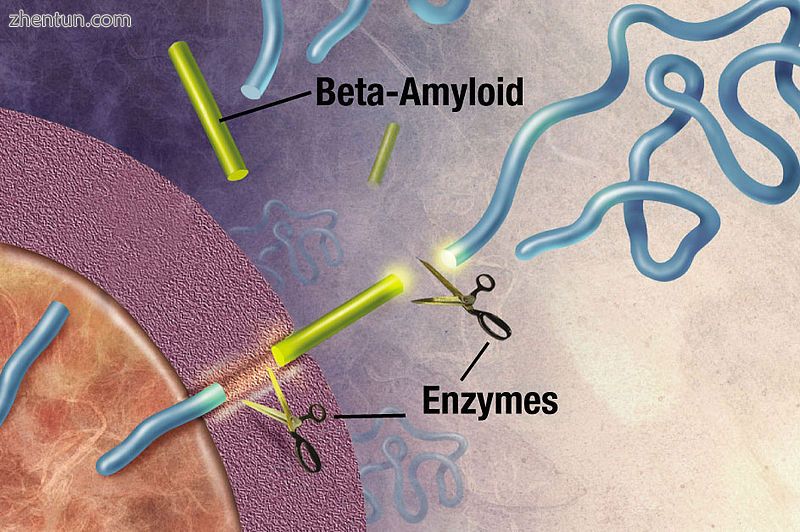
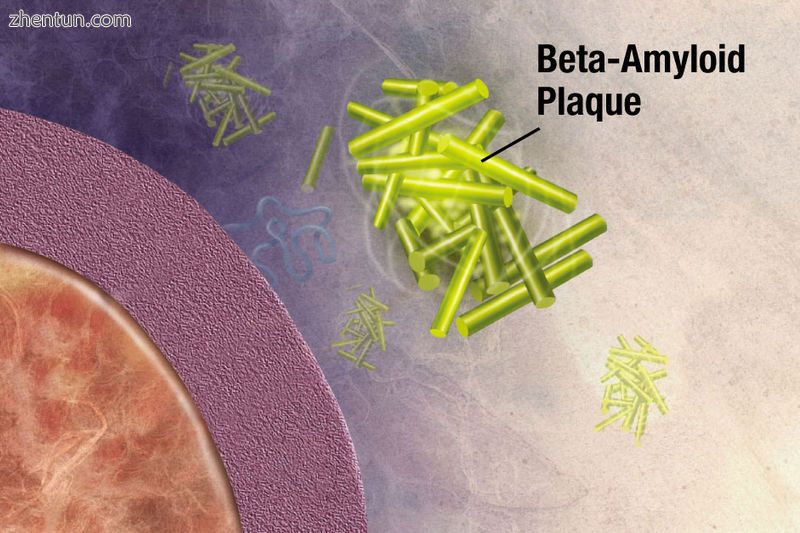
酶作用于APP(淀粉样蛋白前体蛋白)并将其切成片段。 β-淀粉样蛋白片段在AD中形成老年斑是至关重要的。
阿尔茨海默病已被确定为蛋白质错误折叠疾病(蛋白病),由大脑中异常折叠的淀粉样蛋白β蛋白和tau蛋白的斑块积聚引起。[99]斑块由长度为39-43个氨基酸的小肽组成,称为β淀粉样蛋白(Aβ)。 Aβ是来自较大的淀粉样蛋白前体蛋白(APP)的片段。 APP是一种穿透神经元膜的跨膜蛋白。 APP对神经元的生长,存活和伤后修复至关重要。[100] [101]在阿尔茨海默病中,γ分泌酶和β分泌酶在蛋白水解过程中共同作用,导致APP被分成较小的片段。[102]这些碎片中的一个产生淀粉样蛋白β的原纤维,然后形成团块,沉积在称为老年斑的致密形态的神经元外部。[96] [103]
由于tau蛋白的异常聚集,AD也被认为是tau蛋白病。每个神经元都有一个细胞骨架,一个内部支撑结构,部分由称为微管的结构组成。这些微管的作用类似于轨道,将营养物和分子从细胞体引导至轴突和背部的末端。称为tau的蛋白质在磷酸化时稳定微管,因此称为微管相关蛋白。在AD中,tau经历化学变化,变得过度磷酸化;然后它开始与其他线程配对,产生神经原纤维缠结并破坏神经元的运输系统。[104]致病性tau也可通过转座因子失调导致神经元死亡。[105]
疾病机制
确切地说,β-淀粉样肽的产生和聚集的干扰如何引起AD的病理学尚不清楚。[106] [107]淀粉样蛋白假说传统上指的是β-淀粉样蛋白肽的积累作为触发神经元变性的中心事件。聚集的淀粉样蛋白原纤维的累积,被认为是负责破坏细胞钙离子稳态的蛋白质的毒性形式,诱导程序性细胞死亡(细胞凋亡)。[108]还已知Aβ选择性地在阿尔茨海默氏病影响的脑细胞中的线粒体中积累,并且它还抑制某些酶功能和神经元对葡萄糖的利用。[109]
各种炎症过程和细胞因子也可能在阿尔茨海默病的病理学中起作用。 炎症是任何疾病中组织损伤的一般标志物,可能是继发于AD组织损伤或免疫反应的标志物[110]。 越来越多的证据表明神经元与大脑中的免疫机制之间存在强烈的相互作用。 肥胖和全身性炎症可能会干扰促进疾病进展的免疫过程。[111]
在AD [112] [113]中已经描述了不同神经营养因子的分布和它们的受体如脑源性神经营养因子(BDNF)的表达的改变。
诊断
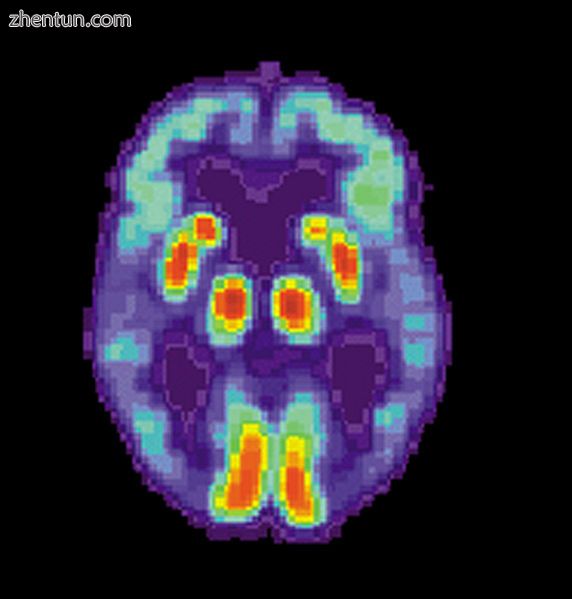
AD患者大脑的PET扫描显示颞叶功能丧失
阿尔茨海默病通常根据患者的病史,亲属的历史和行为观察来诊断。特征性神经和神经心理学特征的存在以及缺乏替代条件是支持性的。[114] [115]先进的医学成像与计算机断层扫描(CT)或磁共振成像(MRI),单光子发射计算机断层扫描(SPECT)或正电子发射断层扫描(PET)可用于帮助排除其他脑病理或痴呆亚型。[ 116]此外,它可以预测从前驱期(轻度认知障碍)到阿尔茨海默病的转变。[117]
包括记忆测试在内的智力功能评估可以进一步描述疾病的状态。[22]医疗机构已经创建了诊断标准,以简化和标准化执业医师的诊断过程。当脑部材料可用时,可以通过非常高的准确性确认诊断,并且可以进行组织学检查。[118]
标准
国家神经和交流障碍与中风研究所(NINCDS)和阿尔茨海默病及相关疾病协会(ADRDA,现称阿尔茨海默氏症协会)于1984年建立了最常用的NINCDS-ADRDA阿尔茨海默氏症诊断标准,[118]于2007年更新。[119]这些标准要求通过神经心理学测试证实存在认知障碍和疑似痴呆综合征,以便对可能或可能的AD进行临床诊断。对于明确的诊断,需要进行组织病理学确认,包括显微镜检查脑组织。在诊断标准和确定的组织病理学确认之间已经显示出良好的统计可靠性和有效性。[120]八个知识领域最常见于AD记忆,语言,感知技能,注意力,运动技能,方向,解决问题和执行功能能力。这些域等同于美国精神病学协会出版的“精神障碍诊断和统计手册”(DSM-IV-TR)中列出的NINCDS-ADRDA阿尔茨海默氏症标准。[121] [122]
技术

神经心理学筛查测试可以帮助诊断AD。在测试中,人们被要求复制类似于图片中所示的图纸,记住单词,读取和减去序列号。
诸如迷你精神状态检查(MMSE)的神经心理学测试被广泛用于评估诊断所需的认知障碍。更全面的测试阵列对于高可靠性结果是必要的,特别是在疾病的早期阶段。[123] [124] AD早期的神经系统检查通常会提供正常结果,除了明显的认知障碍,这可能与其他疾病过程(包括痴呆的其他原因)产生的认知障碍没有差别。
进一步的神经系统检查对AD和其他疾病的鉴别诊断至关重要。[22]对家庭成员的访谈也用于评估疾病。看护人可以提供关于日常生活能力的重要信息,以及随着时间的推移,人的心理功能的减少。[125]看护人的观点尤其重要,因为患有AD的人通常不知道自己的缺陷。[126]很多时候,家庭在检测初期痴呆症状方面也有困难,可能无法向医生传达准确的信息。[127]
补充测试提供有关疾病某些特征的额外信息,或用于排除其他诊断。血液检查可以确定痴呆症的其他原因而不是AD [22] - 可能在极少数情况下是可逆的。[128]通常进行甲状腺功能检查,评估B12,排除梅毒,排除代谢问题(包括肾功能,电解质水平和糖尿病的检测),评估重金属(如铅,汞)和贫血的水平。 (还有必要排除谵妄)。
抑郁症的心理测试被采用,因为抑郁症可能与AD同时发生(见阿尔茨海默病的抑郁症),认知障碍的早期征兆,[129]甚至原因[130] [131]。
由于准确性较低,当人们出现轻度认知障碍(MCI)迹象时,不建议将C-PIB-PET扫描用作早期诊断工具或预测阿尔茨海默病的发展。[132]使用⁸⁸F-FDG PET扫描作为单一测试来识别可能患有阿尔茨海默病的人也没有证据支持。[133]
预防

虽然没有找到因果关系,但在流行病学研究中,下棋等智力活动与定期社交互动的知识活动有关,降低了AD的风险。
没有明确的证据支持任何特定措施有效预防AD。[13]关于预防或延迟AD发病的措施的全球研究往往产生不一致的结果。流行病学研究提出了某些可改变因素之间的关系,如饮食,心血管风险,药品或智力活动等,以及人群患AD的可能性。只有进一步的研究,包括临床试验,才能揭示这些因素是否有助于预防AD。[13]
药物治疗
虽然高胆固醇血症,高血压,糖尿病和吸烟等心血管危险因素与发病风险和AD病程相关,[134] [135]他汀类药物,即降胆固醇药物,在预防或预防方面无效。改善疾病的进程。[136] [137] [138]
2007年认为长期使用非甾体类抗炎药(NSAIDs)与发生AD的可能性降低有关[139]。证据还表明,NSAIDs可以减少与淀粉样蛋白斑相关的炎症,但由于高度不良事件,试验暂停。[13]没有完成预防试验。[13]它们似乎没有用作治疗方法,但截至2011年被认为是候选药物作为症状前预防药。[140]更年期激素替代疗法虽然以前使用过,但可能会增加痴呆症的风险。[141]
生活方式
从事阅读,玩棋盘游戏,填写填字游戏,演奏乐器或定期进行社交互动等智力活动的人士可以降低患阿尔茨海默病的风险。[142]这与认知储备理论是一致的,该理论指出,一些生活经历会导致更有效的神经功能,为个体提供延迟痴呆症表现的认知储备。[142]教育会延迟AD综合症的发作,而不会改变疾病的持续时间。[143]即使在以后的生活中学习第二语言似乎也会延迟患阿尔茨海默病。[144]身体活动也与降低AD风险有关。[143]体育锻炼与痴呆症发病率下降有关。[145]体育锻炼对于减少阿尔茨海默病患者的症状严重程度也是有效的。[146]
饮食
保持健康,日本或地中海饮食的人降低了患AD的风险。[147]地中海饮食可以改善患有这种疾病的人的预后。[148]那些食用富含饱和脂肪和简单碳水化合物(单糖和二糖)的人的风险较高。[149]已经提出地中海饮食的有益心血管作用是作用机制。[150]
关于膳食成分的结论有时难以确定,因为基于人群的研究和随机对照试验的结果不同。[147]有限的证据表明轻度至中度使用酒精,特别是红酒,与较低的AD风险有关。[147]有初步证据表明咖啡因可能具有保护作用。[151]许多富含类黄酮的食物,如可可,红酒和茶,可以降低患AD的风险。[152] [153]
关于使用维生素和矿物质的评论尚未找到足够的一致证据来推荐它们。这包括维生素A,[154] [155] C,[156] [157]α-生育酚形式的维生素E,[158]硒,[159]锌,[160] [161]和叶酸有或没有维生素B12。[162]来自一项随机对照试验的证据表明,维生素E的α-生育酚形式可能会减缓认知能力下降,这一证据被认为质量“适中”。[158]检查叶酸(B9)和其他B族维生素的试验未能显示与认知能力下降有任何显着关联。[163]来自植物和鱼类以及膳食二十二碳六烯酸(DHA)的Omega-3脂肪酸补充剂似乎对患有轻度至中度阿尔茨海默病的人没有益处。[164] [165]
尽管动物有暂定证据,但截至2010年的姜黄素并未显示出对人的益处。[166]有证据表明银杏对认知障碍和痴呆症有任何积极影响。[167]截至2008年,没有具体证据表明大麻素能有效改善AD或痴呆症的症状; [168]然而,对内源性大麻素的一些研究看起来很有希望。[169]
管理
阿尔茨海默病无法治愈; 可用的治疗方法提供相对较小的症状益处,但仍然是姑息性的。 目前的治疗方法可分为药物治疗,社会心理治疗和护理治疗。
药物
多奈哌齐的三维分子模型,用于治疗AD症状的乙酰胆碱酯酶抑制剂

美金刚的分子结构,一种批准用于晚期AD症状的药物
目前有五种药物用于治疗AD的认知问题:四种是乙酰胆碱酯酶抑制剂(他克林,利凡斯的明,加兰他敏和多奈哌齐),另一种(美金刚)是NMDA受体拮抗剂。使用它们的好处很小。[6] [170] [171]没有明确的药物可以延迟或阻止疾病的进展。
降低胆碱能神经元的活性是阿尔茨海默病的一个众所周知的特征。[172]乙酰胆碱酯酶抑制剂用于降低乙酰胆碱(ACh)分解的速度,从而增加脑中ACh的浓度,并对抗由胆碱能神经元死亡引起的ACh损失。[173]有证据表明这些药物对轻度至中度阿尔茨海默病有效,[174] [171] [170]以及一些证据表明它们在晚期使用[170]。这些药物在轻度认知障碍中的使用对延迟AD发病没有任何影响。[175]最常见的副作用是恶心和呕吐,两者都与胆碱能过量有关。这些副作用在大约10-20%的使用者中出现,严重程度为轻度至中度,并且可以通过缓慢调整药物剂量来控制。[176]不太常见的继发效应包括肌肉痉挛,心率降低(心动过缓),食欲和体重下降,以及胃酸产生增加。[174]
谷氨酸是神经系统的兴奋性神经递质,尽管大脑中的过量可通过称为兴奋性毒性的过程导致细胞死亡,该过程由谷氨酸受体的过度刺激组成。兴奋性毒性不仅发生在阿尔茨海默病中,还发生在其他神经系统疾病中,如帕金森病和多发性硬化症。[177]美金刚是非竞争性NMDA受体拮抗剂,首先用作抗流感剂。它通过阻断NMDA受体并抑制谷氨酸过度刺激而作用于谷氨酸能系统。[177] [178]美金刚已被证明在治疗阿尔茨海默病方面有一点益处。[179]美金刚报告的不良事件罕见且轻微,包括幻觉,精神错乱,头晕,头痛和疲劳。[180]美金刚和多奈哌齐的组合已被证明“具有统计学意义,但在临床上具有临床效果”。[181]
非典型抗精神病药适用于减少阿尔茨海默病患者的攻击性和精神病,但其优势可被严重的不良反应所抵消,如中风,运动困难或认知能力下降。[182]从长远来看,它们被证明与死亡率增加有关。[183]在这群人中停止使用抗精神病药似乎是安全的。[184]
Huperzine虽然很有希望,但在推荐之前需要进一步的证据。[185]
心理社会干预
心理社会干预被用作药物治疗的辅助手段,可以分为行为,情绪,认知或刺激导向的方法。对功效的研究尚无法获得,而且很少针对AD,因此一般都将重点放在痴呆症上。[186]
行为干预试图识别和减少问题行为的前因和后果。这种方法未能在改善整体功能方面取得成功,[187]但可以帮助减少一些特定的问题行为,例如尿失禁。[188]关于这些技术在其他行为问题(例如游荡)中的有效性缺乏高质量的数据。[189] [190]音乐疗法可有效减少行为和心理症状。[191]
以情绪为导向的干预措施包括回忆疗法,验证疗法,支持性心理疗法,感觉统合,也称为snoezelen,以及模拟存在疗法。 Cochrane评价未发现任何证据表明这是有效的。[192]支持性心理治疗很少或根本没有正式的科学研究,但一些临床医生发现它可以帮助轻度受损的人适应他们的疾病。[186]回忆疗法(RT)涉及单独或分组讨论过去的经历,多次借助于照片,家庭用品,音乐和录音或过去的其他熟悉物品。 2018年对RT有效性的回顾发现,效果不一致,体积小,临床意义可疑,并且因设置而异。[193]模拟存在疗法(SPT)基于依恋理论,并且涉及播放具有阿尔茨海默氏病患者最亲近的亲属的声音的录音。有部分证据表明SPT可能会减少挑战行为。[194]最后,验证疗法的基础是接受另一个人的经验的现实和个人真相,而感觉整合是基于旨在刺激感官的练习。没有证据支持这些疗法的有效性。[195] [196]
认知导向治疗的目标,包括现实取向和认知再训练,是减少认知缺陷。现实取向包括提供有关时间,地点或人的信息,以便于人们了解周围环境及其在其中的位置。另一方面,认知再训练试图通过锻炼心理能力来改善受损能力。两者都显示出一些改善认知能力的功效,[197] [198]尽管在一些研究中这些影响是短暂的,但也有报道称挫折等负面影响。[186]
以刺激为导向的治疗方法包括艺术,音乐和宠物疗法,运动以及任何其他类型的娱乐活动。刺激对改善行为,情绪以及在较小程度上改善功能有适度的支持。然而,与这些影响同样重要的是,使用刺激疗法的主要支持是人的日常生活中的变化。[186] AD中非侵入性脑刺激和侵入性脑刺激的功效仍然不确定。[199]
看护
进一步的信息:照顾和痴呆症
由于阿尔茨海默氏症无法治愈,并且逐渐使人无法照顾自己的需要,因此护理基本上是治疗,必须在疾病过程中谨慎管理。
在早期和中期阶段,改变生活环境和生活方式可以提高患者安全性并减少看护人的负担。[200] [201]这种修改的例子包括遵守简化的程序,放置安全锁,标记家居用品以提示患有疾病的人或使用改良的日常生活物品。[186] [202] [203]如果进食成为问题,食物需要以较小的碎片或甚至是泥制成。[204]当吞咽困难出现时,可能需要使用喂食管。在这种情况下,持续喂养的医疗功效和道德规范是护理人员和家庭成员的重要考虑因素。[205] [206]在疾病的任何阶段都很少使用身体束缚,尽管有些情况下它们是必要的,以防止对AD患者或其护理人员造成伤害。[186]
随着疾病的进展,可能出现不同的医学问题,例如口腔和牙齿疾病,压力性溃疡,营养不良,卫生问题,或呼吸,皮肤或眼部感染。谨慎的管理可以防止他们,而当他们出现时需要专业的治疗。[207] [208]在疾病的最后阶段,治疗的重点是缓解直至死亡的不适,通常是在临终关怀的帮助下。[209]
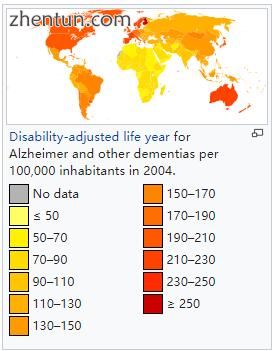
预测
2004年,每10万居民中阿尔茨海默病和其他痴呆症的残疾调整生命年。
阿尔茨海默病的早期阶段很难诊断。一旦认知障碍影响日常生活活动,通常会做出明确的诊断,尽管这个人可能仍然独立生活。这些症状将从轻微的认知问题发展,例如记忆丧失,通过认知和非认知障碍的增加阶段,消除任何独立生活的可能性,特别是在疾病的晚期阶段。[29]
AD患者的预期寿命降低。[210]诊断后,通常为3至10年。[210]
只有不到3%的人生活超过14年。[211]与存活率降低显着相关的疾病特征是认知障碍的严重性增加,功能水平降低,跌倒史和神经学检查中的紊乱。其他一致的疾病,如心脏病,糖尿病或酒精滥用史也与生存期缩短有关。[212] [213] [214]虽然发病年龄越早,总生存年数越高,与年轻人的健康人群相比,预期寿命会特别降低。[215]男性的生存预后不如女性。[211] [216]
肺炎和脱水是AD引起的最常见的直接死亡原因,而癌症死亡的频率低于一般人群。[216]
流行病学
65岁以后的[217]
![Rates after age 65[217].jpg Rates after age 65[217].jpg](data/attachment/forum/201904/28/103359fs2nhmns4lbsi7lc.jpg)
流行病学研究中使用了两种主要措施:发病率和患病率。发病率是每单位人员处于风险中的新病例数(通常为每千人年新病例数);而患病率是指在任何特定时间内人群中疾病病例的总数。
关于发病率,队列纵向研究(多年来一直关注无病人群的研究)为所有痴呆症提供10至15‰人年 - 和AD 5-为5-8,,[217] [218]这意味着每年有一半的新痴呆病例是AD。年龄增长是该疾病的主要危险因素,所有年龄段的发病率并不相同:65岁以后每五年,患病风险大约翻了一番,从3人增加到每千人年69人。 。[217] [218]发病率也存在性别差异,女性患AD的风险较高,尤其是85岁以上的人群。[218] [219]在美国,非西班牙裔白种人死于阿尔茨海默病的风险比非西班牙裔黑人人群高26%,而西班牙裔人口的风险比非西班牙裔白种人低30%。 [220]
由于包括阿尔茨海默病在内的痴呆症,2012年每百万人死亡

人群中AD的患病率取决于不同因素,包括发病率和存活率。由于AD的发病率随着年龄的增长而增加,因此包括感兴趣群体的平均年龄尤为重要。在美国,2000年阿尔茨海默氏症患病率在整体和65-74岁年龄组中估计为1.6%,75-84组的患病率增加到19%,而84-84组的患病率增加到42%。 [221]欠发达地区的患病率较低。[222]世界卫生组织估计,2005年全世界有0.379%的人患有痴呆症,2015年的患病率将增加到0.441%,到2030年将增加到0.556%。[223]其他研究也得出了类似的结论。[222]另一项研究估计,2006年,世界人口的0.40%(范围0.17-0.89%;绝对数量2660万,范围11.4-59.4百万)受到AD的折磨,流行率将增加三倍,绝对数量2050将翻两番。 [224]
历史
另见:阿尔茨海默病的时间表

Alois Alzheimer的病人Auguste Deter于1902年.Hers是第一个被称为阿尔茨海默病的病例。
古希腊和罗马的哲学家和医生将老年与痴呆症联系起来。[18]直到1901年,德国精神病学家Alois Alzheimer才发现了第一例阿尔茨海默病,以他的名字命名,在一名五十岁的女性中,他称之为奥古斯特·D。他跟踪她的病例,直到她于1906年去世。他首先公开报道。[225]在接下来的五年中,医学文献报道了11例类似病例,其中一些已经使用了阿尔茨海默病这一术语。[18]在抑制Auguste D的原始报告中包含的一些临床(妄想和幻觉)和病理特征(动脉硬化改变)后,该疾病首先被Emil Kraepelin描述为一种独特的疾病。[226]他将阿尔茨海默病(也称为Kraepelin的早老性痴呆症)列为1910年7月15日出版的第八版“精神病学教科书”中的老年性痴呆亚型。[227]
在20世纪的大部分时间里,阿尔茨海默病的诊断是针对年龄在45到65岁之间出现痴呆症症状的个体。 1977年,当AD会议得出结论,早老性和老年痴呆症的临床和病理表现几乎相同时,术语发生了变化,尽管作者还补充说,这并不排除他们有不同原因的可能性。[228]这最终导致阿尔茨海默病的诊断与年龄无关。[229]阿尔茨海默型老年痴呆症(SDAT)一词用于描述65岁以上患者的病情,经典阿尔茨海默病用于描述年轻人。最终,术语阿尔茨海默病在医学术语中被正式采用,以描述具有特征性共同症状模式,疾病过程和神经病理学的所有年龄段的个体。[230]
社会与文化
另见:阿尔茨海默病组织
社会成本
老年痴呆症,特别是阿尔茨海默病,可能是欧洲和美国社会上最昂贵的疾病之一,[19] [20]而其他国家如阿根廷,[231]和韩国[232]的成本是也很高涨。随着社会的老龄化,这些成本可能会增加,成为一个重要的社会问题。与AD相关的费用包括直接医疗费用,如养老院护理,直接非医疗费用,如家庭日托,以及间接费用,如患者和护理人员的生产力损失。[20]研究之间的数字不尽相同,但全世界的痴呆症费用计算在1600亿美元左右[233],而美国阿尔茨海默病的费用每年可能达到1000亿美元。[20]
社会成本的最大起源是医疗保健专业人员的长期护理,特别是制度化,这相当于社会总成本的2/3。[19]在家中的生活费用也很高,[19]特别是考虑到家庭的非正式费用,例如看护时间和照顾者的收入损失。[234]
成本随着痴呆症严重程度和行为障碍的存在而增加[235],并与提供身体护理所需的护理时间增加有关。[234]因此,任何减缓认知能力下降,延迟制度化或减少护理人员工作时间的治疗都将带来经济效益。对目前治疗方法的经济评估显示出积极的结果。[20]
护理负担
进一步的信息:照顾和痴呆症
主要照顾者的角色通常由配偶或近亲承担。[236]阿尔茨海默氏症以给予照顾者很大的负担而闻名,包括社会,心理,身体或经济方面。[14] [237] [238] AD患者及其家人通常更喜欢家庭护理。[239]这一选择还延迟或消除了对更专业和更昂贵的护理水平的需求。[239] [240]然而,三分之二的养老院居民患有痴呆症。[186]
痴呆症护理人员的身体和精神障碍率很高。[241]与主要照顾者的更大社会心理问题相关的因素包括在家中有受影响的人,照顾者是配偶,要求受照顾者的行为,如抑郁,行为紊乱,幻觉,睡眠问题或行走中断和社会孤立。[242 ] [243]关于经济问题,家庭照顾者经常放弃工作时间,平均每周花费47小时与AD患者一起,而照顾他们的费用很高。根据研究,在美国,照顾阿尔茨海默病患者的直接和间接费用平均每年在18,000美元至77,500美元之间。[234] [236]
认知行为疗法和单独或集体应对策略的教学已经证明了它们在改善看护者心理健康方面的功效。[14] [244]
媒体
主要文章:媒体中的阿尔茨海默病
AD已在电影中被描绘成:Iris(2001),基于John Bayley的妻子Iris Murdoch的回忆录; [245] The Notebook(2004),基于Nicholas Sparks的1996年同名小说; [246] A要记住的时刻(2004); Thanmathra(2005); [247]明日的回忆(Ashita no Kioku)(2006),基于Hiroshi Ogiwara的同名小说; [248]远离她(2006),基于爱丽丝Munro的短篇小说“The Bear Came over the Mountain”; [249] Still Alice(2014),关于哥伦比亚大学教授,早发性阿尔茨海默病,基于Lisa Genova 2007年的同名小说,并以Julianne Moore为主题角色。阿尔茨海默病的纪录片包括Malcolm和Barbara:一个爱情故事(1999)和Malcolm and Barbara:Love's Farewell(2007),两者都以Malcolm Pointon为特色。[250] [251] [252]
研究方向
主要文章:阿尔茨海默病的研究
药物治疗
在2002 - 2012年期间,在第一阶段,第二阶段或第三阶段试验中评估了244种化合物,其中只有一种(美金刚)获得了FDA批准(尽管其他化合物仍处于试验阶段)。[253] Solanezumab和aducanumab未能显示已患有阿尔茨海默症症状的人的有效性。[254]
临床研究的一个领域是关注治疗潜在的疾病病理学。 β-淀粉样蛋白水平的降低是研究中化合物[255](例如阿扑吗啡)的常见目标。淀粉样蛋白的免疫疗法或疫苗接种是研究中的一种治疗方式。[256]与预防性疫苗接种不同,推定的治疗方法将用于治疗已经诊断出的人。它基于训练免疫系统识别,攻击和逆转淀粉样蛋白沉积的概念,从而改变疾病的进程。[257]正在调查的此类疫苗的一个例子是ACC-001,[258] [259],尽管试验于2008年暂停。[260]另一种类似的药物是bapineuzumab,一种与天然诱导的抗淀粉样蛋白抗体相同的抗体。[261]然而,已发现免疫治疗剂会引起一些有关的药物不良反应,例如淀粉样蛋白相关的影像异常。[262]其他方法是神经保护剂,如AL-108,[263]和金属 - 蛋白质相互作用衰减剂,如PBT2。[264]一种TNFα受体阻断融合蛋白依那西普显示出令人鼓舞的结果。[265]
2008年,两项独立的临床试验显示,用氯化甲硫堇(一种抑制tau聚集的药物)[266] [267]和Dimebon(一种抗组胺药)改变轻度至中度AD的病程有积极效果。[268] Dimebon的连续III期试验未能在主要和次要终点显示出积极效果。[269] [270] [271]使用甲基硫堇氯化物表明,甲基硫堇从肠道的生物利用度受到摄食和胃酸的影响,导致意外变化的剂量。[272]作为前药LMTX的新稳定制剂正处于III期试验阶段(2014年)。[273]
行为预防
关于冥想对检索记忆和认知功能的影响的初步研究令人鼓舞。[274] [合格证据] 2015年的一项评论表明,基于正念的干预措施可以预防或延缓轻度认知障碍和阿尔茨海默病的发作。[275]
可能的传输
目前正在研究人与人之间可能传播的罕见病例,[276]例如生长激素患者。[277]
感染
已发现单纯疱疹病毒HSV-1与淀粉样斑块相同。[278]这表明AD可以用抗病毒药物治疗或预防。[278] [279]对细胞培养中的抗病毒药物的研究已显示出有希望的结果。[280]
还描述了AD脑的真菌感染。[281]这一假设是由微生物学家L. Carrasco提出的,当时他的研究小组发现播散性真菌病与AD之间存在统计学相关性。[282]进一步的研究表明,AD患者的不同脑区存在真菌感染,而对照组则不然。[283] [284]真菌感染解释了AD患者中观察到的症状。 AD的缓慢进展符合某些全身性真菌感染的慢性特征,这些感染可能是无症状的,因此未被注意和未经治疗。[283]真菌假说也与一些其他既定的AD假设相容,如淀粉样蛋白假说,可以解释为免疫系统对中枢神经系统感染的反应,[285] [286] [287]如R. Moir和R. Tanzi在AD的小鼠和蠕虫模型中。
成像
在许多可用的医学成像技术中,单光子发射计算机断层扫描(SPECT)似乎在区分阿尔茨海默病与其他类型的痴呆症方面具有优势,并且与心理测试和病史分析相比,这已被证明具有更高的准确性。 [288]进展导致了新诊断标准的提议。[22] [119]
PiB PET仍在研究中,但类似的PET扫描放射性药物称为florbetapir,含有较长时间的放射性核素氟-18,是阿尔茨海默病的诊断工具。[289] [290]
淀粉样蛋白成像可能与其他标记物一起使用,而不是作为替代物。[291]体积MRI可以检测大脑区域大小的变化。测量在阿尔茨海默病进展期间萎缩的那些区域作为诊断指标显示出前景。它可能比目前正在研究的其他成像方法更便宜。[292]
2011年,FDA小组一致投票建议批准florbetapir。成像剂可以帮助检测阿尔茨海默氏症的脑斑。[293]阴性扫描表明稀疏或无斑块,这与AD的诊断不一致。[294]
诊断
阿尔茨海默氏症的研究重点放在症状开始前诊断病情。[295]已经开发了许多生化测试以实现早期检测。一些此类测试涉及脑脊液分析β-淀粉样蛋白,总tau蛋白和磷酸化tau181P蛋白浓度。[296]因为绘制CSF会很痛苦,所以避免重复绘制。循环miRNA和炎症生物标志物的血液检测是一种潜在的替代指标。[296]
参考
Burns A, Iliffe S (February 2009). "Alzheimer's disease". BMJ. 338: b158. doi:10.1136/bmj.b158. PMID 19196745.
"Dementia Fact sheet". World Health Organization. 12 December 2017.
Mendez MF (November 2012). "Early-onset Alzheimer's disease: nonamnestic subtypes and type 2 AD". Archives of Medical Research. 43 (8): 677–85. doi:10.1016/j.arcmed.2012.11.009. PMC 3532551. PMID 23178565.
Ballard C, Gauthier S, Corbett A, Brayne C, Aarsland D, Jones E (March 2011). "Alzheimer's disease". Lancet. 377 (9770): 1019–31. doi:10.1016/S0140-6736(10)61349-9. PMID 21371747.
"Dementia diagnosis and assessment" (PDF). National Institute for Health and Care Excellence (NICE). Archived from the original (PDF) on 5 December 2014. Retrieved 30 November 2014.
Commission de la transparence (June 2012). "Drugs for Alzheimer's disease: best avoided. No therapeutic advantage" [Drugs for Alzheimer's disease: best avoided. No therapeutic advantage]. Prescrire International. 21 (128): 150. PMID 22822592.
Querfurth HW, LaFerla FM (January 2010). "Alzheimer's disease". The New England Journal of Medicine. 362 (4): 329–44. doi:10.1056/NEJMra0909142. PMID 20107219.
GBD 2015 Disease Injury Incidence Prevalence Collaborators (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
GBD 2015 Mortality Causes of Death Collaborators (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/S0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
"About Alzheimer's Disease: Symptoms". National Institute on Aging. Archived from the original on 15 January 2012. Retrieved 28 December 2011.
Todd S, Barr S, Roberts M, Passmore AP (November 2013). "Survival in dementia and predictors of mortality: a review". International Journal of Geriatric Psychiatry. 28 (11): 1109–24. doi:10.1002/gps.3946. PMID 23526458.
"So, What Can You Do?". National Institute on Aging. 29 July 2016. Archived from the original on 3 April 2017.
Hsu D, Marshall GA (2017). "Primary and Secondary Prevention Trials in Alzheimer Disease: Looking Back, Moving Forward". Current Alzheimer Research. 14 (4): 426–440. doi:10.2174/1567205013666160930112125. PMC 5329133. PMID 27697063.
Thompson CA, Spilsbury K, Hall J, Birks Y, Barnes C, Adamson J (July 2007). "Systematic review of information and support interventions for caregivers of people with dementia". BMC Geriatrics. 7: 18. doi:10.1186/1471-2318-7-18. PMC 1951962. PMID 17662119.
Forbes D, Forbes SC, Blake CM, Thiessen EJ, Forbes S (April 2015). "Exercise programs for people with dementia". The Cochrane Database of Systematic Reviews (Submitted manuscript). 132 (4): CD006489. doi:10.1002/14651858.CD006489.pub4. PMID 25874613.
National Institute for Health and Clinical Excellence. "Low-dose antipsychotics in people with dementia". National Institute for Health and Care Excellence (NICE). Archived from the original on 5 December 2014. Retrieved 29 November 2014.
"Information for Healthcare Professionals: Conventional Antipsychotics". US Food and Drug Administration. 16 June 2008. Archived from the original on 29 November 2014. Retrieved 29 November 2014.
Berchtold NC, Cotman CW (1998). "Evolution in the conceptualization of dementia and Alzheimer's disease: Greco-Roman period to the 1960s". Neurobiology of Aging. 19 (3): 173–89. doi:10.1016/S0197-4580(98)00052-9. PMID 9661992.
Bonin-Guillaume S, Zekry D, Giacobini E, Gold G, Michel JP (January 2005). "[The economical impact of dementia]". Presse Médicale (in French). 34 (1): 35–41. doi:10.1016/s0755-4982(05)83882-5. PMID 15685097.
Meek PD, McKeithan K, Schumock GT (1998). "Economic considerations in Alzheimer's disease". Pharmacotherapy. 18 (2 Pt 2): 68–73, discussion 79–82. doi:10.1002/j.1875-9114.1998.tb03880.x (inactive 24 February 2019). PMID 9543467.
"Evaluating Prescription Drugs Used to Treat: Alzheimer's Disease Comparing Effectiveness, Safety, and Price" (PDF). Consumer Reports Drug Effectiveness Review Project. Consumer Reports. May 2012. Archived (PDF) from the original on 5 September 2012. Retrieved 1 May 2013.
Waldemar G, Dubois B, Emre M, Georges J, McKeith IG, Rossor M, Scheltens P, Tariska P, Winblad B (January 2007). "Recommendations for the diagnosis and management of Alzheimer's disease and other disorders associated with dementia: EFNS guideline". European Journal of Neurology. 14 (1): e1–26. doi:10.1111/j.1468-1331.2006.01605.x. PMID 17222085.
Bäckman L, Jones S, Berger AK, Laukka EJ, Small BJ (September 2004). "Multiple cognitive deficits during the transition to Alzheimer's disease". Journal of Internal Medicine. 256 (3): 195–204. doi:10.1111/j.1365-2796.2004.01386.x. PMID 15324363.
Nygård L (2003). "Instrumental activities of daily living: a stepping-stone towards Alzheimer's disease diagnosis in subjects with mild cognitive impairment?". Acta Neurologica Scandinavica. Supplementum. 179 (179): 42–6. doi:10.1034/j.1600-0404.107.s179.8.x. PMID 12603250.
Arnáiz E, Almkvist O (2003). "Neuropsychological features of mild cognitive impairment and preclinical Alzheimer's disease". Acta Neurologica Scandinavica. Supplementum. 179: 34–41. doi:10.1034/j.1600-0404.107.s179.7.x. PMID 12603249.
Landes AM, Sperry SD, Strauss ME, Geldmacher DS (December 2001). "Apathy in Alzheimer's disease". Journal of the American Geriatrics Society. 49 (12): 1700–7. doi:10.1046/j.1532-5415.2001.49282.x. PMID 11844006.
Murray ED, Buttner N, Price BH (2012). "Depression and Psychosis in Neurological Practice". In Bradley WG, Daroff RB, Fenichel GM, Jankovic J (eds.). Bradley's neurology in clinical practice (6th ed.). Philadelphia, PA: Elsevier/Saunders. ISBN 978-1-4377-0434-1.
Grundman M, Petersen RC, Ferris SH, Thomas RG, Aisen PS, Bennett DA, et al. (January 2004). "Mild cognitive impairment can be distinguished from Alzheimer disease and normal aging for clinical trials". Archives of Neurology. 61 (1): 59–66. doi:10.1001/archneur.61.1.59. PMID 14732621.
Förstl H, Kurz A (1999). "Clinical features of Alzheimer's disease". European Archives of Psychiatry and Clinical Neuroscience. 249 (6): 288–90. doi:10.1007/s004060050101. PMID 10653284.
Carlesimo GA, Oscar-Berman M (June 1992). "Memory deficits in Alzheimer's patients: a comprehensive review". Neuropsychology Review. 3 (2): 119–69. doi:10.1007/BF01108841. PMID 1300219.
Jelicic M, Bonebakker AE, Bonke B (1995). "Implicit memory performance of patients with Alzheimer's disease: a brief review". International Psychogeriatrics. 7 (3): 385–92. doi:10.1017/S1041610295002134. PMID 8821346.
Taler V, Phillips NA (July 2008). "Language performance in Alzheimer's disease and mild cognitive impairment: a comparative review". Journal of Clinical and Experimental Neuropsychology. 30 (5): 501–56. doi:10.1080/13803390701550128. PMID 18569251.
Frank EM (September 1994). "Effect of Alzheimer's disease on communication function". Journal of the South Carolina Medical Association. 90 (9): 417–23. PMID 7967534.
Volicer L, Harper DG, Manning BC, Goldstein R, Satlin A (May 2001). "Sundowning and circadian rhythms in Alzheimer's disease". The American Journal of Psychiatry. 158 (5): 704–11. doi:10.1176/appi.ajp.158.5.704. PMID 11329390.
Gold DP, Reis MF, Markiewicz D, Andres D (January 1995). "When home caregiving ends: a longitudinal study of outcomes for caregivers of relatives with dementia". Journal of the American Geriatrics Society. 43 (1): 10–6. doi:10.1111/j.1532-5415.1995.tb06235.x. PMID 7806732.
"What We Know Today About Alzheimer's Disease". Alzheimer's Association. Archived from the original on 7 October 2011. Retrieved 1 October 2011. While scientists know Alzheimer's disease involves progressive brain cell failure, the reason cells fail isn't clear.
Reitz C, Mayeux R (April 2014). "Alzheimer disease: epidemiology, diagnostic criteria, risk factors and biomarkers". Biochemical Pharmacology. 88 (4): 640–51. doi:10.1016/j.bcp.2013.12.024. PMC 3992261. PMID 24398425.
Wilson RS, Barral S, Lee JH, Leurgans SE, Foroud TM, Sweet RA, Graff-Radford N, Bird TD, Mayeux R, Bennett DA (2011). "Heritability of different forms of memory in the Late Onset Alzheimer's Disease Family Study". Journal of Alzheimer's Disease. 23 (2): 249–55. doi:10.3233/JAD-2010-101515. PMC 3130303. PMID 20930268.
Gatz M, Reynolds CA, Fratiglioni L, Johansson B, Mortimer JA, Berg S, Fiske A, Pedersen NL (February 2006). "Role of genes and environments for explaining Alzheimer disease". Archives of General Psychiatry. 63 (2): 168–74. doi:10.1001/archpsyc.63.2.168. PMID 16461860.[unreliable medical source?]
Blennow K, de Leon MJ, Zetterberg H (July 2006). "Alzheimer's disease". Lancet. 368 (9533): 387–403. doi:10.1016/S0140-6736(06)69113-7. PMID 16876668.
Waring SC, Rosenberg RN (March 2008). "Genome-wide association studies in Alzheimer disease". Archives of Neurology. 65 (3): 329–34. doi:10.1001/archneur.65.3.329. PMID 18332245.
Selkoe DJ (June 1999). "Translating cell biology into therapeutic advances in Alzheimer's disease". Nature. 399 (6738 Suppl): A23–31. doi:10.1038/19866. PMID 10392577.
Borchelt DR, Thinakaran G, Eckman CB, Lee MK, Davenport F, Ratovitsky T, et al. (November 1996). "Familial Alzheimer's disease-linked presenilin 1 variants elevate Abeta1-42/1-40 ratio in vitro and in vivo". Neuron. 17 (5): 1005–13. doi:10.1016/S0896-6273(00)80230-5. PMID 8938131.
Shioi J, Georgakopoulos A, Mehta P, Kouchi Z, Litterst CM, Baki L, Robakis NK (May 2007). "FAD mutants unable to increase neurotoxic Abeta 42 suggest that mutation effects on neurodegeneration may be independent of effects on Abeta". Journal of Neurochemistry. 101 (3): 674–81. doi:10.1111/j.1471-4159.2006.04391.x. PMID 17254019.[unreliable medical source?]
Kim, JH (December 2018). "Genetics of Alzheimer's Disease". Dementia and neurocognitive disorders. 17 (4): 131–136. doi:10.12779/dnd.2018.17.4.131. PMID 30906402.
Strittmatter WJ, Saunders AM, Schmechel D, Pericak-Vance M, Enghild J, Salvesen GS, Roses AD (March 1993). "Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease". Proceedings of the National Academy of Sciences of the United States of America. 90 (5): 1977–81. doi:10.1073/pnas.90.5.1977. PMC 46003. PMID 8446617.
Mahley RW, Weisgraber KH, Huang Y (April 2006). "Apolipoprotein E4: a causative factor and therapeutic target in neuropathology, including Alzheimer's disease". Proceedings of the National Academy of Sciences of the United States of America. 103 (15): 5644–51. Bibcode:2006PNAS..103.5644M. doi:10.1073/pnas.0600549103. PMC 1414631. PMID 16567625.
Hall K, Murrell J, Ogunniyi A, Deeg M, Baiyewu O, Gao S, Gureje O, Dickens J, Evans R, Smith-Gamble V, Unverzagt FW, Shen J, Hendrie H (January 2006). "Cholesterol, APOE genotype, and Alzheimer disease: an epidemiologic study of Nigerian Yoruba". Neurology. 66 (2): 223–7. doi:10.1212/01.wnl.0000194507.39504.17. PMC 2860622. PMID 16434658.
Gureje O, Ogunniyi A, Baiyewu O, Price B, Unverzagt FW, Evans RM, et al. (January 2006). "APOE epsilon4 is not associated with Alzheimer's disease in elderly Nigerians". Annals of Neurology. 59 (1): 182–5. doi:10.1002/ana.20694. PMC 2855121. PMID 16278853.
Lambert JC, Ibrahim-Verbaas CA, Harold D, Naj AC, Sims R, Bellenguez C, et al. (December 2013). "Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer's disease". Nature Genetics. 45 (12): 1452–8. doi:10.1038/ng.2802. PMC 3896259. PMID 24162737.
Jonsson T, Stefansson H, Steinberg S, Jonsdottir I, Jonsson PV, Snaedal J, et al. (January 2013). "Variant of TREM2 associated with the risk of Alzheimer's disease". The New England Journal of Medicine (Original article). 368 (2): 107–16. doi:10.1056/NEJMoa1211103. PMC 3677583. PMID 23150908.
Guerreiro R, Wojtas A, Bras J, Carrasquillo M, Rogaeva E, Majounie E, et al. (January 2013). "TREM2 variants in Alzheimer's disease". The New England Journal of Medicine (Original article). 368 (2): 117–27. doi:10.1056/NEJMoa1211851. PMC 3631573. PMID 23150934.
Mukherjee S, Mez J, Trittschuh EH, Saykin AJ, Gibbons LE, Fardo DW, et al. (December 2018). "Genetic data and cognitively defined late-onset Alzheimer's disease subgroups". Molecular Psychiatry. doi:10.1038/s41380-018-0298-8. PMID 30514930.
Francis PT, Palmer AM, Snape M, Wilcock GK (February 1999). "The cholinergic hypothesis of Alzheimer's disease: a review of progress". Journal of Neurology, Neurosurgery, and Psychiatry. 66 (2): 137–47. doi:10.1136/jnnp.66.2.137. PMC 1736202. PMID 10071091.
Martorana A, Esposito Z, Koch G (August 2010). "Beyond the cholinergic hypothesis: do current drugs work in Alzheimer's disease?". CNS Neuroscience & Therapeutics. 16 (4): 235–45. doi:10.1111/j.1755-5949.2010.00175.x. PMID 20560995.
Hardy J, Allsop D (October 1991). "Amyloid deposition as the central event in the aetiology of Alzheimer's disease". Trends in Pharmacological Sciences. 12 (10): 383–8. doi:10.1016/0165-6147(91)90609-V. PMID 1763432.
Mudher A, Lovestone S (January 2002). "Alzheimer's disease-do tauists and baptists finally shake hands?". Trends in Neurosciences. 25 (1): 22–6. doi:10.1016/S0166-2236(00)02031-2. PMID 11801334.
Nistor M, Don M, Parekh M, Sarsoza F, Goodus M, Lopez GE, Kawas C, Leverenz J, Doran E, Lott IT, Hill M, Head E (October 2007). "Alpha- and beta-secretase activity as a function of age and beta-amyloid in Down syndrome and normal brain". Neurobiology of Aging. 28 (10): 1493–506. doi:10.1016/j.neurobiolaging.2006.06.023. PMC 3375834. PMID 16904243.
Lott IT, Head E (March 2005). "Alzheimer disease and Down syndrome: factors in pathogenesis". Neurobiology of Aging. 26 (3): 383–9. doi:10.1016/j.neurobiolaging.2004.08.005. PMID 15639317.
Polvikoski T, Sulkava R, Haltia M, Kainulainen K, Vuorio A, Verkkoniemi A, Niinistö L, Halonen P, Kontula K (November 1995). "Apolipoprotein E, dementia, and cortical deposition of beta-amyloid protein". The New England Journal of Medicine. 333 (19): 1242–7. doi:10.1056/NEJM199511093331902. PMID 7566000.
Transgenic mice:
Games D, Adams D, Alessandrini R, Barbour R, Berthelette P, Blackwell C, Carr T, Clemens J, Donaldson T, Gillespie F (February 1995). "Alzheimer-type neuropathology in transgenic mice overexpressing V717F beta-amyloid precursor protein". Nature. 373 (6514): 523–7. doi:10.1038/373523a0. PMID 7845465.
Masliah E, Sisk A, Mallory M, Mucke L, Schenk D, Games D (September 1996). "Comparison of neurodegenerative pathology in transgenic mice overexpressing V717F beta-amyloid precursor protein and Alzheimer's disease". The Journal of Neuroscience. 16 (18): 5795–811. doi:10.1523/JNEUROSCI.16-18-05795.1996. PMID 8795633.
Hsiao K, Chapman P, Nilsen S, Eckman C, Harigaya Y, Younkin S, Yang F, Cole G (October 1996). "Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice". Science. 274 (5284): 99–102. doi:10.1126/science.274.5284.99. PMID 8810256.
Lalonde R, Dumont M, Staufenbiel M, Sturchler-Pierrat C, Strazielle C (November 2002). "Spatial learning, exploration, anxiety, and motor coordination in female APP23 transgenic mice with the Swedish mutation". Brain Research. 956 (1): 36–44. doi:10.1016/S0006-8993(02)03476-5. PMID 12426044.
Holmes C, Boche D, Wilkinson D, Yadegarfar G, Hopkins V, Bayer A, Jones RW, Bullock R, Love S, Neal JW, Zotova E, Nicoll JA (July 2008). "Long-term effects of Abeta42 immunisation in Alzheimer's disease: follow-up of a randomised, placebo-controlled phase I trial". Lancet. 372 (9634): 216–23. doi:10.1016/S0140-6736(08)61075-2. PMID 18640458.
Lacor PN, Buniel MC, Furlow PW, Clemente AS, Velasco PT, Wood M, et al. (January 2007). "Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer's disease". The Journal of Neuroscience. 27 (4): 796–807. doi:10.1523/JNEUROSCI.3501-06.2007. PMID 17251419.
Laurén J, Gimbel DA, Nygaard HB, Gilbert JW, Strittmatter SM (February 2009). "Cellular prion protein mediates impairment of synaptic plasticity by amyloid-beta oligomers". Nature. 457 (7233): 1128–32. doi:10.1038/nature07761. PMC 2748841. PMID 19242475.
Nikolaev A, McLaughlin T, O'Leary DD, Tessier-Lavigne M (February 2009). "APP binds DR6 to trigger axon pruning and neuron death via distinct caspases". Nature. 457 (7232): 981–9. Bibcode:2009Natur.457..981N. doi:10.1038/nature07767. PMC 2677572. PMID 19225519.
Feuerstein, Adam (14 February 2017). "Merck Alzheimer's Drug Study Halted Early for Futility". New York City, NY, USA: TheStreet, Inc. Archived from the original on 16 February 2017.Merck Alzheimer's Drug Study Halted Early for Futility Independent study monitors concluded that there was "virtually no chance of finding a positive clinical effect."
Goedert M, Spillantini MG, Crowther RA (July 1991). "Tau proteins and neurofibrillary degeneration". Brain Pathology. 1 (4): 279–86. doi:10.1111/j.1750-3639.1991.tb00671.x. PMID 1669718.
Iqbal K, Alonso A, Chen S, Chohan MO, El-Akkad E, Gong CX, Khatoon S, Li B, Liu F, Rahman A, Tanimukai H, Grundke-Iqbal I (January 2005). "Tau pathology in Alzheimer disease and other tauopathies". Biochimica et Biophysica Acta. 1739 (2–3): 198–210. doi:10.1016/j.bbadis.2004.09.008. PMID 15615638.
Chun W, Johnson GV (January 2007). "The role of tau phosphorylation and cleavage in neuronal cell death". Frontiers in Bioscience. 12: 733–56. doi:10.2741/2097. PMID 17127334.
Deane R, Zlokovic BV (April 2007). "Role of the blood-brain barrier in the pathogenesis of Alzheimer's disease". Current Alzheimer Research. 4 (2): 191–7. doi:10.2174/156720507780362245. PMID 17430246.
Xu H, Finkelstein DI, Adlard PA (12 June 2014). "Interactions of metals and Apolipoprotein E in Alzheimer's disease". Frontiers in Aging Neuroscience. 6: 121. doi:10.3389/fnagi.2014.00121. PMC 4054654. PMID 24971061. Although we still do not know if the metal ion dyshomeostasis present in AD is a cause or consequence of the disease, there is a growing body of evidence showing a direct correlation between metal ions and key AD-related key proteins.
Su B, Wang X, Nunomura A, Moreira PI, Lee HG, Perry G, Smith MA, Zhu X (December 2008). "Oxidative stress signaling in Alzheimer's disease". Current Alzheimer Research. 5 (6): 525–32. doi:10.2174/156720508786898451. PMC 2780015. PMID 19075578.
Kastenholz B, Garfin DE, Horst J, Nagel KA (2009). "Plant metal chaperones: a novel perspective in dementia therapy". Amyloid. 16 (2): 81–3. doi:10.1080/13506120902879392. PMID 20536399.
"Aluminium and Alzheimer's disease". Facts about dementia. Alzheimer's Society. Archived from the original on 27 October 2005. Retrieved 14 October 2005.
Bondy SC (January 2016). "Low levels of aluminum can lead to behavioral and morphological changes associated with Alzheimer's disease and age-related neurodegeneration". Neurotoxicology (Submitted manuscript). 52: 222–9. doi:10.1016/j.neuro.2015.12.002. PMID 26687397.
Kandimalla R, Vallamkondu J, Corgiat EB, Gill KD (March 2016). "Understanding Aspects of Aluminum Exposure in Alzheimer's Disease Development". Brain Pathology. 26 (2): 139–54. doi:10.1111/bpa.12333. PMID 26494454.
Santibáñez M, Bolumar F, García AM (November 2007). "Occupational risk factors in Alzheimer's disease: a review assessing the quality of published epidemiological studies". Occupational and Environmental Medicine. 64 (11): 723–32. doi:10.1136/oem.2006.028209. PMC 2078415. PMID 17525096.
Lidsky TI (May 2014). "Is the Aluminum Hypothesis dead?". Journal of Occupational and Environmental Medicine. 56 (5 Suppl): S73–9. doi:10.1097/jom.0000000000000063. PMC 4131942. PMID 24806729.
Yegambaram M, Manivannan B, Beach TG, Halden RU (2015). "Role of environmental contaminants in the etiology of Alzheimer's disease: a review". Current Alzheimer Research. 12 (2): 116–46. doi:10.2174/1567205012666150204121719. PMC 4428475. PMID 25654508.
Cataldo JK, Prochaska JJ, Glantz SA (2010). "Cigarette smoking is a risk factor for Alzheimer's Disease: an analysis controlling for tobacco industry affiliation". Journal of Alzheimer's Disease. 19 (2): 465–80. doi:10.3233/JAD-2010-1240. PMC 2906761. PMID 20110594.
Eikelenboom P, van Exel E, Hoozemans JJ, Veerhuis R, Rozemuller AJ, van Gool WA (2010). "Neuroinflammation - an early event in both the history and pathogenesis of Alzheimer's disease". Neuro-Degenerative Diseases. 7 (1–3): 38–41. doi:10.1159/000283480. PMID 20160456.
Moulton PV, Yang W (2012). "Air pollution, oxidative stress, and Alzheimer's disease". Journal of Environmental and Public Health (Review). 2012: 1–9. doi:10.1155/2012/472751. PMC 3317180. PMID 22523504.
Miklossy J (August 2011). "Alzheimer's disease - a neurospirochetosis. Analysis of the evidence following Koch's and Hill's criteria". Journal of Neuroinflammation. 8 (1): 90. doi:10.1186/1742-2094-8-90. PMC 3171359. PMID 21816039.
Pisa D, Alonso R, Rábano A, Rodal I, Carrasco L (October 2015). "Different Brain Regions are Infected with Fungi in Alzheimer's Disease". Scientific Reports. 5 (1): 15015. Bibcode:2015NatSR...515015P. doi:10.1038/srep15015. PMC 4606562. PMID 26468932.
Bartzokis G (August 2011). "Alzheimer's disease as homeostatic responses to age-related myelin breakdown". Neurobiology of Aging. 32 (8): 1341–71. doi:10.1016/j.neurobiolaging.2009.08.007. PMC 3128664. PMID 19775776.
Cai Z, Xiao M (2016). "Oligodendrocytes and Alzheimer's disease". The International Journal of Neuroscience. 126 (2): 97–104. doi:10.3109/00207454.2015.1025778. PMID 26000818.
Reisberg B, Franssen EH, Hasan SM, Monteiro I, Boksay I, Souren LE, et al. (1999). "Retrogenesis: clinical, physiologic, and pathologic mechanisms in brain aging, Alzheimer's and other dementing processes". European Archives of Psychiatry and Clinical Neuroscience. 249 Suppl 3 (3): 28–36. doi:10.1007/pl00014170. PMID 10654097.
Alves GS, Oertel Knöchel V, Knöchel C, Carvalho AF, Pantel J, Engelhardt E, Laks J (2015). "Integrating retrogenesis theory to Alzheimer's disease pathology: insight from DTI-TBSS investigation of the white matter microstructural integrity". BioMed Research International. 2015: 291658. doi:10.1155/2015/291658. PMC 4320890. PMID 25685779.
Brenner Carson, Verna (2015). Caregiving for Alzheimer's Disease. New York: Springer New York Academy of Sciences. pp. 1–9. ISBN 978-1-4939-2406-6.
Zis, Panagiotis; Hadjivassiliou, Marios (26 February 2019). "Treatment of Neurological Manifestations of Gluten Sensitivity and Coeliac Disease". Current Treatment Options in Neurology. 21 (3). doi:10.1007/s11940-019-0552-7.
Makhlouf S, Messelmani M, Zaouali J, Mrissa R (2018). "Cognitive impairment in celiac disease and non-celiac gluten sensitivity: review of literature on the main cognitive impairments, the imaging and the effect of gluten free diet". Acta Neurol Belg (Review). 118 (1): 21–27. doi:10.1007/s13760-017-0870-z. PMID 29247390.
Wenk GL (2003). "Neuropathologic changes in Alzheimer's disease". The Journal of Clinical Psychiatry. 64 Suppl 9: 7–10. PMID 12934968.
Braak H, Del Tredici K (December 2012). "Where, when, and in what form does sporadic Alzheimer's disease begin?". Current Opinion in Neurology. 25 (6): 708–14. doi:10.1097/WCO.0b013e32835a3432. PMID 23160422.
Desikan RS, Cabral HJ, Hess CP, Dillon WP, Glastonbury CM, Weiner MW, Schmansky NJ, Greve DN, Salat DH, Buckner RL, Fischl B (August 2009). "Automated MRI measures identify individuals with mild cognitive impairment and Alzheimer's disease". Brain. 132 (Pt 8): 2048–57. doi:10.1093/brain/awp123. PMC 2714061. PMID 19460794.
Moan R (20 July 2009). "MRI Software Accurately IDs Preclinical Alzheimer's Disease". Diagnostic Imaging. Archived from the original on 16 May 2016. Retrieved 7 January 2013.
Tiraboschi P, Hansen LA, Thal LJ, Corey-Bloom J (June 2004). "The importance of neuritic plaques and tangles to the development and evolution of AD". Neurology. 62 (11): 1984–9. doi:10.1212/01.WNL.0000129697.01779.0A. PMID 15184601.
Bouras C, Hof PR, Giannakopoulos P, Michel JP, Morrison JH (1994). "Regional distribution of neurofibrillary tangles and senile plaques in the cerebral cortex of elderly patients: a quantitative evaluation of a one-year autopsy population from a geriatric hospital". Cerebral Cortex. 4 (2): 138–50. doi:10.1093/cercor/4.2.138. PMID 8038565.
Kotzbauer PT, Trojanowsk JQ, Lee VM (October 2001). "Lewy body pathology in Alzheimer's disease". Journal of Molecular Neuroscience. 17 (2): 225–32. doi:10.1385/JMN:17:2:225. PMID 11816795.
Hashimoto M, Rockenstein E, Crews L, Masliah E (2003). "Role of protein aggregation in mitochondrial dysfunction and neurodegeneration in Alzheimer's and Parkinson's diseases". Neuromolecular Medicine. 4 (1–2): 21–36. doi:10.1385/NMM:4:1-2:21. PMID 14528050.
Priller C, Bauer T, Mitteregger G, Krebs B, Kretzschmar HA, Herms J (July 2006). "Synapse formation and function is modulated by the amyloid precursor protein". The Journal of Neuroscience. 26 (27): 7212–21. doi:10.1523/JNEUROSCI.1450-06.2006. PMID 16822978.
Turner PR, O'Connor K, Tate WP, Abraham WC (May 2003). "Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory". Progress in Neurobiology. 70 (1): 1–32. doi:10.1016/S0301-0082(03)00089-3. PMID 12927332.
Hooper NM (April 2005). "Roles of proteolysis and lipid rafts in the processing of the amyloid precursor protein and prion protein". Biochemical Society Transactions. 33 (Pt 2): 335–8. doi:10.1042/BST0330335. PMID 15787600.
Ohnishi S, Takano K (March 2004). "Amyloid fibrils from the viewpoint of protein folding". Cellular and Molecular Life Sciences. 61 (5): 511–524. doi:10.1007/s00018-003-3264-8. PMID 15004691.
Hernández F, Avila J (September 2007). "Tauopathies". Cellular and Molecular Life Sciences. 64 (17): 2219–33. doi:10.1007/s00018-007-7220-x. PMID 17604998.
Sun W, Samimi H, Gamez M, Zare H, Frost B (August 2018). "Pathogenic tau-induced piRNA depletion promotes neuronal death through transposable element dysregulation in neurodegenerative tauopathies". Nature Neuroscience. 21 (8): 1038–1048. doi:10.1038/s41593-018-0194-1. PMC 6095477. PMID 30038280.
Van Broeck B, Van Broeckhoven C, Kumar-Singh S (2007). "Current insights into molecular mechanisms of Alzheimer disease and their implications for therapeutic approaches". Neuro-Degenerative Diseases. 4 (5): 349–65. doi:10.1159/000105156. PMID 17622778.
Huang Y, Mucke L (March 2012). "Alzheimer mechanisms and therapeutic strategies". Cell. 148 (6): 1204–22. doi:10.1016/j.cell.2012.02.040. PMC 3319071. PMID 22424230.
Yankner BA, Duffy LK, Kirschner DA (October 1990). "Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides". Science. 250 (4978): 279–82. Bibcode:1990Sci...250..279Y. doi:10.1126/science.2218531. PMID 2218531.
Chen X, Yan SD (December 2006). "Mitochondrial Abeta: a potential cause of metabolic dysfunction in Alzheimer's disease". IUBMB Life. 58 (12): 686–94. doi:10.1080/15216540601047767. PMID 17424907.
Greig NH, Mattson MP, Perry T, Chan SL, Giordano T, Sambamurti K, Rogers JT, Ovadia H, Lahiri DK (December 2004). "New therapeutic strategies and drug candidates for neurodegenerative diseases: p53 and TNF-alpha inhibitors, and GLP-1 receptor agonists". Annals of the New York Academy of Sciences. 1035: 290–315. doi:10.1196/annals.1332.018. PMID 15681814.
Heneka MT, Carson MJ, El Khoury J, Landreth GE, Brosseron F, Feinstein DL, et al. (April 2015). "Neuroinflammation in Alzheimer's disease". The Lancet. Neurology. 14 (4): 388–405. doi:10.1016/S1474-4422(15)70016-5. PMC 5909703. PMID 25792098.
Tapia-Arancibia L, Aliaga E, Silhol M, Arancibia S (November 2008). "New insights into brain BDNF function in normal aging and Alzheimer disease". Brain Research Reviews. 59 (1): 201–20. doi:10.1016/j.brainresrev.2008.07.007. PMID 18708092.
Schindowski K, Belarbi K, Buée L (February 2008). "Neurotrophic factors in Alzheimer's disease: role of axonal transport". Genes, Brain, and Behavior. 7 (Suppl 1): 43–56. doi:10.1111/j.1601-183X.2007.00378.x. PMC 2228393. PMID 18184369.
Mendez MF (2006). "The accurate diagnosis of early-onset dementia". International Journal of Psychiatry in Medicine. 36 (4): 401–12. doi:10.2190/Q6J4-R143-P630-KW41. PMID 17407994.
Klafki HW, Staufenbiel M, Kornhuber J, Wiltfang J (November 2006). "Therapeutic approaches to Alzheimer's disease". Brain. 129 (Pt 11): 2840–55. doi:10.1093/brain/awl280. PMID 17018549.
Dementia: Quick Reference Guide (PDF). London: (UK) National Institute for Health and Clinical Excellence. November 2006. ISBN 978-1-84629-312-2. Archived from the original (PDF) on 27 February 2008. Retrieved 22 February 2008.
Schroeter ML, Stein T, Maslowski N, Neumann J (October 2009). "Neural correlates of Alzheimer's disease and mild cognitive impairment: a systematic and quantitative meta-analysis involving 1351 patients". NeuroImage. 47 (4): 1196–206. doi:10.1016/j.neuroimage.2009.05.037. PMC 2730171. PMID 19463961.
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (July 1984). "Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease". Neurology. 34 (7): 939–44. doi:10.1212/wnl.34.7.939. PMID 6610841.
Dubois B, Feldman HH, Jacova C, Dekosky ST, Barberger-Gateau P, Cummings J, et al. (August 2007). "Research criteria for the diagnosis of Alzheimer's disease: revising the NINCDS-ADRDA criteria". The Lancet. Neurology. 6 (8): 734–46. doi:10.1016/S1474-4422(07)70178-3. PMID 17616482.
Blacker D, Albert MS, Bassett SS, Go RC, Harrell LE, Folstein MF (December 1994). "Reliability and validity of NINCDS-ADRDA criteria for Alzheimer's disease. The National Institute of Mental Health Genetics Initiative". Archives of Neurology. 51 (12): 1198–204. doi:10.1001/archneur.1994.00540240042014. PMID 7986174.
American Psychiatric Association (2000). Diagnostic and statistical manual of mental disorders: DSM-IV-TR (4th Edition Text Revision ed.). Washington, DC: American Psychiatric Association. ISBN 978-0-89042-025-6.
Ito N (May 1996). "[Clinical aspects of dementia]". [Hokkaido Igaku Zasshi] the Hokkaido Journal of Medical Science (in Japanese). 71 (3): 315–20. PMID 8752526.
Tombaugh TN, McIntyre NJ (September 1992). "The mini-mental state examination: a comprehensive review". Journal of the American Geriatrics Society. 40 (9): 922–35. doi:10.1111/j.1532-5415.1992.tb01992.x. PMID 1512391.
Pasquier F (January 1999). "Early diagnosis of dementia: neuropsychology". Journal of Neurology. 246 (1): 6–15. doi:10.1007/s004150050299. PMID 9987708.
Harvey PD, Moriarty PJ, Kleinman L, Coyne K, Sadowsky CH, Chen M, Mirski DF (2005). "The validation of a caregiver assessment of dementia: the Dementia Severity Scale". Alzheimer Disease and Associated Disorders. 19 (4): 186–94. doi:10.1097/01.wad.0000189034.43203.60. PMID 16327345.
Antoine C, Antoine P, Guermonprez P, Frigard B (2004). "[Awareness of deficits and anosognosia in Alzheimer's disease]". L'Encephale (in French). 30 (6): 570–7. doi:10.1016/S0013-7006(04)95472-3. PMID 15738860.
Cruz VT, Pais J, Teixeira A, Nunes B (2004). "[The initial symptoms of Alzheimer disease: caregiver perception]". Acta Medica Portuguesa (in Portuguese). 17 (6): 435–44. PMID 16197855.
Clarfield AM (October 2003). "The decreasing prevalence of reversible dementias: an updated meta-analysis". Archives of Internal Medicine. 163 (18): 2219–29. doi:10.1001/archinte.163.18.2219. PMID 14557220.
Sun X, Steffens DC, Au R, Folstein M, Summergrad P, Yee J, Rosenberg I, Mwamburi DM, Qiu WQ (May 2008). "Amyloid-associated depression: a prodromal depression of Alzheimer disease?". Archives of General Psychiatry. 65 (5): 542–50. doi:10.1001/archpsyc.65.5.542. PMC 3042807. PMID 18458206.
Geldmacher DS, Whitehouse PJ (May 1997). "Differential diagnosis of Alzheimer's disease". Neurology. 48 (5 Suppl 6): S2–9. doi:10.1212/WNL.48.5_Suppl_6.2S. PMID 9153154.
Potter GG, Steffens DC (May 2007). "Contribution of depression to cognitive impairment and dementia in older adults". The Neurologist. 13 (3): 105–17. doi:10.1097/01.nrl.0000252947.15389.a9. PMID 17495754.
Zhang S, Smailagic N, Hyde C, Noel-Storr AH, Takwoingi Y, McShane R, Feng J (July 2014). "(11)C-PIB-PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI)". The Cochrane Database of Systematic Reviews (7): CD010386. doi:10.1002/14651858.CD010386.pub2. PMID 25052054.
Smailagic N, Vacante M, Hyde C, Martin S, Ukoumunne O, Sachpekidis C (January 2015). "1⁸F-FDG PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI)". The Cochrane Database of Systematic Reviews. 1: CD010632. doi:10.1002/14651858.CD010632.pub2. PMID 25629415.
Patterson C, Feightner JW, Garcia A, Hsiung GY, MacKnight C, Sadovnick AD (February 2008). "Diagnosis and treatment of dementia: 1. Risk assessment and primary prevention of Alzheimer disease". CMAJ. 178 (5): 548–56. doi:10.1503/cmaj.070796. PMC 2244657. PMID 18299540.
Rosendorff C, Beeri MS, Silverman JM (2007). "Cardiovascular risk factors for Alzheimer's disease". The American Journal of Geriatric Cardiology. 16 (3): 143–9. doi:10.1111/j.1076-7460.2007.06696.x. PMID 17483665.
Reiss AB, Wirkowski E (2007). "Role of HMG-CoA reductase inhibitors in neurological disorders : progress to date". Drugs. 67 (15): 2111–20. doi:10.2165/00003495-200767150-00001. PMID 17927279.
Kuller LH (August 2007). "Statins and dementia". Current Atherosclerosis Reports. 9 (2): 154–61. doi:10.1007/s11883-007-0012-9. PMID 17877925.
McGuinness B, Craig D, Bullock R, Malouf R, Passmore P (July 2014). "Statins for the treatment of dementia". The Cochrane Database of Systematic Reviews. 7 (7): CD007514. doi:10.1002/14651858.CD007514.pub3. PMID 25004278.
Szekely CA, Town T, Zandi PP (2007). NSAIDs for the chemoprevention of Alzheimer's disease. Sub-Cellular Biochemistry. Subcellular Biochemistry. 42. pp. 229–48. doi:10.1007/1-4020-5688-5_11. ISBN 978-1-4020-5687-1. PMID 17612054.
Hoozemans JJ, Veerhuis R, Rozemuller JM, Eikelenboom P (February 2011). "Soothing the inflamed brain: effect of non-steroidal anti-inflammatory drugs on Alzheimer's disease pathology". CNS & Neurological Disorders Drug Targets. 10 (1): 57–67. doi:10.2174/187152711794488665. PMID 21143138.
Marjoribanks J, Farquhar C, Roberts H, Lethaby A, Lee J (January 2017). "Long-term hormone therapy for perimenopausal and postmenopausal women". The Cochrane Database of Systematic Reviews. 1: CD004143. doi:10.1002/14651858.CD004143.pub5. PMID 28093732.
Stern Y (July 2006). "Cognitive reserve and Alzheimer disease". Alzheimer Disease and Associated Disorders. 20 (3 Suppl 2): S69–74. doi:10.1097/01.wad.0000213815.20177.19. PMID 16917199.
Paradise M, Cooper C, Livingston G (February 2009). "Systematic review of the effect of education on survival in Alzheimer's disease". International Psychogeriatrics. 21 (1): 25–32. doi:10.1017/S1041610208008053. PMID 19026089.
Neergaard, Lauran (19 February 2011). "Speaking 2 Languages May Delay Getting Alzheimer's". The Denver Post. Associated Press. Archived from the original on 2 May 2014.
Cheng ST (September 2016). "Cognitive Reserve and the Prevention of Dementia: the Role of Physical and Cognitive Activities". Current Psychiatry Reports. 18 (9): 85. doi:10.1007/s11920-016-0721-2. PMC 4969323. PMID 27481112.
Farina N, Rusted J, Tabet N (January 2014). "The effect of exercise interventions on cognitive outcome in Alzheimer's disease: a systematic review". International Psychogeriatrics. 26 (1): 9–18. doi:10.1017/S1041610213001385. PMID 23962667.
Hu N, Yu JT, Tan L, Wang YL, Sun L, Tan L (2013). "Nutrition and the risk of Alzheimer's disease". BioMed Research International (Review). 2013: 1–12. doi:10.1155/2013/524820. PMC 3705810. PMID 23865055.
Solfrizzi V, Panza F, Frisardi V, Seripa D, Logroscino G, Imbimbo BP, Pilotto A (May 2011). "Diet and Alzheimer's disease risk factors or prevention: the current evidence". Expert Review of Neurotherapeutics. 11 (5): 677–708. doi:10.1586/ern.11.56. PMID 21539488.
Kanoski SE, Davidson TL (April 2011). "Western diet consumption and cognitive impairment: links to hippocampal dysfunction and obesity". Physiology & Behavior (Review). 103 (1): 59–68. doi:10.1016/j.physbeh.2010.12.003. PMC 3056912. PMID 21167850.
Solfrizzi V, Capurso C, D'Introno A, Colacicco AM, Santamato A, Ranieri M, Fiore P, Capurso A, Panza F (January 2008). "Lifestyle-related factors in predementia and dementia syndromes". Expert Review of Neurotherapeutics. 8 (1): 133–58. doi:10.1586/14737175.8.1.133. PMID 18088206.
Santos C, Costa J, Santos J, Vaz-Carneiro A, Lunet N (2010). "Caffeine intake and dementia: systematic review and meta-analysis". Journal of Alzheimer's Disease. 20 Suppl 1: S187–204. doi:10.3233/JAD-2010-091387. PMID 20182026.
Nehlig A (March 2013). "The neuroprotective effects of cocoa flavanol and its influence on cognitive performance". British Journal of Clinical Pharmacology (Review). 75 (3): 716–27. doi:10.1111/j.1365-2125.2012.04378.x. PMC 3575938. PMID 22775434.
Stoclet JC, Schini-Kerth V (March 2011). "[Dietary flavonoids and human health]". Annales Pharmaceutiques Francaises. 69 (2): 78–90. doi:10.1016/j.pharma.2010.11.004. PMID 21440100.
Ono K, Yamada M (April 2012). "Vitamin A and Alzheimer's disease". Geriatrics & Gerontology International (Review). 12 (2): 180–8. doi:10.1111/j.1447-0594.2011.00786.x. PMID 22221326.
Lerner AJ, Gustaw-Rothenberg K, Smyth S, Casadesus G (March – April 2012). "Retinoids for treatment of Alzheimer's disease". BioFactors. 38 (2): 84–9. doi:10.1002/biof.196. PMID 22419567.
Heo JH, Lee KM (March 2013). "The possible role of antioxidant vitamin C in Alzheimer's disease treatment and prevention". American Journal of Alzheimer's Disease and Other Dementias (Review). 28 (2): 120–5. doi:10.1177/1533317512473193. PMID 23307795.
Boothby LA, Doering PL (December 2005). "Vitamin C and vitamin E for Alzheimer's disease". The Annals of Pharmacotherapy. 39 (12): 2073–80. doi:10.1345/aph.1E495. PMID 16227450.
Farina N, Llewellyn D, Isaac MG, Tabet N (April 2017). "Vitamin E for Alzheimer's dementia and mild cognitive impairment". The Cochrane Database of Systematic Reviews. 4: CD002854. doi:10.1002/14651858.CD002854.pub5. PMID 28418065.
Loef M, Schrauzer GN, Walach H (2011). "Selenium and Alzheimer's disease: a systematic review". Journal of Alzheimer's Disease (Review). 26 (1): 81–104. doi:10.3233/JAD-2011-110414. PMID 21593562.
Loef M, von Stillfried N, Walach H (September 2012). "Zinc diet and Alzheimer's disease: a systematic review". Nutritional Neuroscience (Review). 15 (5): 2–12. doi:10.1179/1476830512Y.0000000010. PMID 22583839.
Avan A, Hoogenraad TU (2015). "Zinc and Copper in Alzheimer's Disease". Journal of Alzheimer's Disease (Review). 46 (1): 89–92. doi:10.3233/JAD-150186. PMID 25835420.
Malouf R, Grimley Evans J (October 2008). "Folic acid with or without vitamin B12 for the prevention and treatment of healthy elderly and demented people". The Cochrane Database of Systematic Reviews (4): CD004514. doi:10.1002/14651858.CD004514.pub2. PMID 18843658.
Wald DS, Kasturiratne A, Simmonds M (June 2010). "Effect of folic acid, with or without other B vitamins, on cognitive decline: meta-analysis of randomized trials". The American Journal of Medicine. 123 (6): 522–527.e2. doi:10.1016/j.amjmed.2010.01.017. PMID 20569758.
Cunnane SC, Chouinard-Watkins R, Castellano CA, Barberger-Gateau P (January 2013). "Docosahexaenoic acid homeostasis, brain aging and Alzheimer's disease: Can we reconcile the evidence?". Prostaglandins, Leukotrienes, and Essential Fatty Acids. 88 (1): 61–70. doi:10.1016/j.plefa.2012.04.006. PMID 22575581.
Burckhardt M, Herke M, Wustmann T, Watzke S, Langer G, Fink A (April 2016). "Omega-3 fatty acids for the treatment of dementia". The Cochrane Database of Systematic Reviews. 4: CD009002. doi:10.1002/14651858.CD009002.pub3. PMID 27063583.
Hamaguchi T, Ono K, Yamada M (October 2010). "REVIEW: Curcumin and Alzheimer's disease". CNS Neuroscience & Therapeutics (review). 16 (5): 285–97. doi:10.1111/j.1755-5949.2010.00147.x. PMID 20406252.
Birks J, Grimley Evans J (January 2009). "Ginkgo biloba for cognitive impairment and dementia". The Cochrane Database of Systematic Reviews (1): CD003120. doi:10.1002/14651858.CD003120.pub3. PMID 19160216.
Krishnan S, Cairns R, Howard R (April 2009). Krishnan S (ed.). "Cannabinoids for the treatment of dementia". The Cochrane Database of Systematic Reviews (2): CD007204. doi:10.1002/14651858.CD007204.pub2. PMID 19370677.
Bilkei-Gorzo A (December 2012). "The endocannabinoid system in normal and pathological brain ageing". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 367 (1607): 3326–41. doi:10.1098/rstb.2011.0388. PMC 3481530. PMID 23108550.
Birks JS, Harvey RJ (June 2018). "Donepezil for dementia due to Alzheimer's disease". The Cochrane Database of Systematic Reviews. 6: CD001190. doi:10.1002/14651858.CD001190.pub3. PMID 29923184.
Birks JS, Grimley Evans J (April 2015). "Rivastigmine for Alzheimer's disease". The Cochrane Database of Systematic Reviews (4): CD001191. doi:10.1002/14651858.CD001191.pub3. PMID 25858345.
Geula C, Mesulam MM (1995). "Cholinesterases and the pathology of Alzheimer disease". Alzheimer Disease and Associated Disorders. 9 Suppl 2: 23–8. doi:10.1097/00002093-199501002-00005. PMID 8534419.
Stahl SM (November 2000). "The new cholinesterase inhibitors for Alzheimer's disease, Part 2: illustrating their mechanisms of action". The Journal of Clinical Psychiatry. 61 (11): 813–4. doi:10.4088/JCP.v61n1101. PMID 11105732.
Birks J (January 2006). Birks J (ed.). "Cholinesterase inhibitors for Alzheimer's disease". The Cochrane Database of Systematic Reviews (1): CD005593. doi:10.1002/14651858.CD005593. PMID 16437532.
Raschetti R, Albanese E, Vanacore N, Maggini M (November 2007). "Cholinesterase inhibitors in mild cognitive impairment: a systematic review of randomised trials". PLoS Medicine. 4 (11): e338. doi:10.1371/journal.pmed.0040338. PMC 2082649. PMID 18044984.
al.], edited by Brian K. Alldredge ... [et (2013). Applied therapeutics : the clinical use of drugs (10th ed.). Baltimore: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 2385. ISBN 978-1609137137.
Lipton SA (February 2006). "Paradigm shift in neuroprotection by NMDA receptor blockade: memantine and beyond". Nature Reviews. Drug Discovery. 5 (2): 160–70. doi:10.1038/nrd1958. PMID 16424917.
"Memantine". US National Library of Medicine (Medline). 4 January 2004. Archived from the original on 22 February 2010. Retrieved 3 February 2010.
McShane R, Areosa Sastre A, Minakaran N (April 2006). "Memantine for dementia". The Cochrane Database of Systematic Reviews (2): CD003154. doi:10.1002/14651858.CD003154.pub5. PMID 16625572.
"Namenda prescribing information" (PDF). Forest Pharmaceuticals. Archived from the original (PDF) on 27 February 2008. Retrieved 19 February 2008. (primary source)
Raina P, Santaguida P, Ismaila A, Patterson C, Cowan D, Levine M, Booker L, Oremus M (March 2008). "Effectiveness of cholinesterase inhibitors and memantine for treating dementia: evidence review for a clinical practice guideline". Annals of Internal Medicine. 148 (5): 379–97. doi:10.7326/0003-4819-148-5-200803040-00009. PMID 18316756.
Ballard C, Waite J (January 2006). Ballard CG (ed.). "The effectiveness of atypical antipsychotics for the treatment of aggression and psychosis in Alzheimer's disease". The Cochrane Database of Systematic Reviews (1): CD003476. doi:10.1002/14651858.CD003476.pub2. PMID 16437455.
Ballard C, Hanney ML, Theodoulou M, Douglas S, McShane R, Kossakowski K, Gill R, Juszczak E, Yu LM, Jacoby R (February 2009). "The dementia antipsychotic withdrawal trial (DART-AD): long-term follow-up of a randomised placebo-controlled trial". The Lancet. Neurology. 8 (2): 151–7. doi:10.1016/S1474-4422(08)70295-3. PMID 19138567. Lay summary.
Declercq T, Petrovic M, Azermai M, Vander Stichele R, De Sutter AI, van Driel ML, Christiaens T (March 2013). "Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia". The Cochrane Database of Systematic Reviews. 3 (3): CD007726. doi:10.1002/14651858.CD007726.pub2. hdl:1854/LU-3109108. PMID 23543555.
Li J, Wu HM, Zhou RL, Liu GJ, Dong BR (April 2008). "Huperzine A for Alzheimer's disease". The Cochrane Database of Systematic Reviews (2): CD005592. doi:10.1002/14651858.CD005592.pub2. PMID 18425924.
Rabins PV, Blacker D, Rovner BW, Rummans T, Schneider LS, Tariot PN, et al. (Steering Committee on Practice Guidelines) (December 2007). "American Psychiatric Association practice guideline for the treatment of patients with Alzheimer's disease and other dementias. Second edition". The American Journal of Psychiatry. 164 (12 Suppl): 5–56. PMID 18340692.
Bottino CM, Carvalho IA, Alvarez AM, Avila R, Zukauskas PR, Bustamante SE, et al. (December 2005). "Cognitive rehabilitation combined with drug treatment in Alzheimer's disease patients: a pilot study". Clinical Rehabilitation. 19 (8): 861–9. doi:10.1191/0269215505cr911oa. PMID 16323385.
Doody RS, Stevens JC, Beck C, Dubinsky RM, Kaye JA, Gwyther L, et al. (May 2001). "Practice parameter: management of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology". Neurology. 56 (9): 1154–66. doi:10.1212/WNL.56.9.1154. PMID 11342679.
Hermans DG, Htay UH, McShane R (January 2007). "Non-pharmacological interventions for wandering of people with dementia in the domestic setting". The Cochrane Database of Systematic Reviews (1): CD005994. doi:10.1002/14651858.CD005994.pub2. PMID 17253573.
Robinson L, Hutchings D, Dickinson HO, Corner L, Beyer F, Finch T, Hughes J, Vanoli A, Ballard C, Bond J (January 2007). "Effectiveness and acceptability of non-pharmacological interventions to reduce wandering in dementia: a systematic review". International Journal of Geriatric Psychiatry. 22 (1): 9–22. doi:10.1002/gps.1643. PMID 17096455.
Abraha I, Rimland JM, Trotta FM, Dell'Aquila G, Cruz-Jentoft A, Petrovic M, Gudmundsson A, Soiza R, O'Mahony D, Guaita A, Cherubini A (March 2017). "Systematic review of systematic reviews of non-pharmacological interventions to treat behavioural disturbances in older patients with dementia. The SENATOR-OnTop series". BMJ Open. 7 (3): e012759. doi:10.1136/bmjopen-2016-012759. PMC 5372076. PMID 28302633.
Chung JC, Lai CK, Chung PM, French HP (2002). "Snoezelen for dementia". The Cochrane Database of Systematic Reviews (4): CD003152. doi:10.1002/14651858.CD003152. PMID 12519587.
Woods B, O'Philbin L, Farrell EM, Spector AE, Orrell M (March 2018). "Reminiscence therapy for dementia". The Cochrane Database of Systematic Reviews. 3: CD001120. doi:10.1002/14651858.CD001120.pub3. PMID 29493789.
Zetteler J (November 2008). "Effectiveness of simulated presence therapy for individuals with dementia: a systematic review and meta-analysis". Aging & Mental Health. 12 (6): 779–85. doi:10.1080/13607860802380631. PMID 19023729.
Neal M, Barton Wright P (2003). Neal M (ed.). "Validation therapy for dementia". The Cochrane Database of Systematic Reviews (3): CD001394. doi:10.1002/14651858.CD001394. PMID 12917907.
Chung JC, Lai CK, Chung PM, French HP (2002). Chung JC (ed.). "Snoezelen for dementia". The Cochrane Database of Systematic Reviews (4): CD003152. doi:10.1002/14651858.CD003152. PMID 12519587. (up to date as of 2009)
Spector A, Orrell M, Davies S, Woods B (July 2007). Spector AE (ed.). "WITHDRAWN: Reality orientation for dementia". The Cochrane Database of Systematic Reviews (3): CD001119. doi:10.1002/14651858.CD001119.pub2. PMID 17636652.
Spector A, Thorgrimsen L, Woods B, Royan L, Davies S, Butterworth M, Orrell M (September 2003). "Efficacy of an evidence-based cognitive stimulation therapy programme for people with dementia: randomised controlled trial". The British Journal of Psychiatry. 183 (3): 248–54. doi:10.1192/bjp.183.3.248. PMID 12948999.
Chang CH, Lane HY, Lin CH (2018). "Brain Stimulation in Alzheimer's Disease". Frontiers in Psychiatry. 9: 201. doi:10.3389/fpsyt.2018.00201. PMC 5992378. PMID 29910746. This article incorporates text by Chun-Hung Chang, Hsien-Yuan Lane, and Chieh-Hsin Lin available under the CC BY 4.0 license.
Gitlin LN, Corcoran M, Winter L, Boyce A, Hauck WW (February 2001). "A randomized, controlled trial of a home environmental intervention: effect on efficacy and upset in caregivers and on daily function of persons with dementia". The Gerontologist. 41 (1): 4–14. doi:10.1093/geront/41.1.4. PMID 11220813.
Gitlin LN, Hauck WW, Dennis MP, Winter L (March 2005). "Maintenance of effects of the home environmental skill-building program for family caregivers and individuals with Alzheimer's disease and related disorders". The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences. 60 (3): 368–74. doi:10.1093/gerona/60.3.368. PMID 15860476.
"Treating Behavioral and Psychiatric Symptoms". Alzheimer's Association. 2006. Archived from the original on 25 September 2006. Retrieved 25 September 2006.
Dunne TE, Neargarder SA, Cipolloni PB, Cronin-Golomb A (August 2004). "Visual contrast enhances food and liquid intake in advanced Alzheimer's disease". Clinical Nutrition. 23 (4): 533–8. doi:10.1016/j.clnu.2003.09.015. PMID 15297089.
Dudek SG (2007). Nutrition Essentials for Nursing Practice. Hagerstown, Maryland: Lippincott Williams & Wilkins. p. 360. ISBN 978-0-7817-6651-7. Retrieved 19 August 2008.
Dennehy C (2006). "Analysis of patients' rights: dementia and PEG insertion". British Journal of Nursing. 15 (1): 18–20. doi:10.12968/bjon.2006.15.1.20303. PMID 16415742.
Chernoff R (April 2006). "Tube feeding patients with dementia". Nutrition in Clinical Practice. 21 (2): 142–6. doi:10.1177/0115426506021002142. PMID 16556924.
Gambassi G, Landi F, Lapane KL, Sgadari A, Mor V, Bernabei R (July 1999). "Predictors of mortality in patients with Alzheimer's disease living in nursing homes". Journal of Neurology, Neurosurgery, and Psychiatry. 67 (1): 59–65. doi:10.1136/jnnp.67.1.59. PMC 1736445. PMID 10369823.
Medical issues:
Head B (January 2003). "Palliative care for persons with dementia". Home Healthcare Nurse. 21 (1): 53–60, quiz 61. doi:10.1097/00004045-200301000-00012. PMID 12544465.
Friedlander AH, Norman DC, Mahler ME, Norman KM, Yagiela JA (September 2006). "Alzheimer's disease: psychopathology, medical management and dental implications". Journal of the American Dental Association. 137 (9): 1240–51. doi:10.14219/jada.archive.2006.0381. PMID 16946428.
Belmin J (2007). "Practical guidelines for the diagnosis and management of weight loss in Alzheimer's disease: a consensus from appropriateness ratings of a large expert panel". The Journal of Nutrition, Health & Aging. 11 (1): 33–7. PMID 17315078.
McCurry SM, Gibbons LE, Logsdon RG, Vitiello M, Teri L (October 2003). "Training caregivers to change the sleep hygiene practices of patients with dementia: the NITE-AD project". Journal of the American Geriatrics Society. 51 (10): 1455–60. doi:10.1046/j.1532-5415.2003.51466.x. PMID 14511168.
Perls TT, Herget M (December 1995). "Higher respiratory infection rates on an Alzheimer's special care unit and successful intervention". Journal of the American Geriatrics Society. 43 (12): 1341–4. doi:10.1111/j.1532-5415.1995.tb06611.x. PMID 7490383.
Shega JW, Levin A, Hougham GW, Cox-Hayley D, Luchins D, Hanrahan P, Stocking C, Sachs GA (April 2003). "Palliative Excellence in Alzheimer Care Efforts (PEACE): a program description". Journal of Palliative Medicine. 6 (2): 315–20. doi:10.1089/109662103764978641. PMID 12854952.
Zanetti O, Solerte SB, Cantoni F (2009). "Life expectancy in Alzheimer's disease (AD)". Archives of Gerontology and Geriatrics. 49 Suppl 1: 237–43. doi:10.1016/j.archger.2009.09.035. PMID 19836639.
Mölsä PK, Marttila RJ, Rinne UK (March 1995). "Long-term survival and predictors of mortality in Alzheimer's disease and multi-infarct dementia". Acta Neurologica Scandinavica. 91 (3): 159–64. doi:10.1111/j.1600-0404.1995.tb00426.x. PMID 7793228.
Bowen JD, Malter AD, Sheppard L, Kukull WA, McCormick WC, Teri L, Larson EB (August 1996). "Predictors of mortality in patients diagnosed with probable Alzheimer's disease". Neurology. 47 (2): 433–9. doi:10.1212/wnl.47.2.433. PMID 8757016.
Larson EB, Shadlen MF, Wang L, McCormick WC, Bowen JD, Teri L, Kukull WA (April 2004). "Survival after initial diagnosis of Alzheimer disease". Annals of Internal Medicine. 140 (7): 501–9. doi:10.7326/0003-4819-140-7-200404060-00008. PMID 15068977.
Jagger C, Clarke M, Stone A (January 1995). "Predictors of survival with Alzheimer's disease: a community-based study". Psychological Medicine. 25 (1): 171–7. doi:10.1017/S0033291700028191. PMID 7792352.
Dodge HH, Shen C, Pandav R, DeKosky ST, Ganguli M (February 2003). "Functional transitions and active life expectancy associated with Alzheimer disease". Archives of Neurology. 60 (2): 253–9. doi:10.1001/archneur.60.2.253. PMID 12580712.
Ganguli M, Dodge HH, Shen C, Pandav RS, DeKosky ST (May 2005). "Alzheimer disease and mortality: a 15-year epidemiological study". Archives of Neurology. 62 (5): 779–84. doi:10.1001/archneur.62.5.779. PMID 15883266.
Bermejo-Pareja F, Benito-León J, Vega S, Medrano MJ, Román GC (January 2008). "Incidence and subtypes of dementia in three elderly populations of central Spain". Journal of the Neurological Sciences. 264 (1–2): 63–72. doi:10.1016/j.jns.2007.07.021. PMID 17727890.
Di Carlo A, Baldereschi M, Amaducci L, Lepore V, Bracco L, Maggi S, Bonaiuto S, Perissinotto E, Scarlato G, Farchi G, Inzitari D (January 2002). "Incidence of dementia, Alzheimer's disease, and vascular dementia in Italy. The ILSA Study". Journal of the American Geriatrics Society. 50 (1): 41–8. doi:10.1046/j.1532-5415.2002.50006.x. PMID 12028245.
Andersen K, Launer LJ, Dewey ME, Letenneur L, Ott A, Copeland JR, et al. (December 1999). "Gender differences in the incidence of AD and vascular dementia: The EURODEM Studies. EURODEM Incidence Research Group". Neurology. 53 (9): 1992–7. doi:10.1212/wnl.53.9.1992. PMID 10599770.
Tejada-Vera B. (2013). Mortality from Alzheimer's Disease in the United States: Data for 2000 and 2010. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
2000 U.S. estimates:
Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA (August 2003). "Alzheimer disease in the US population: prevalence estimates using the 2000 census". Archives of Neurology. 60 (8): 1119–22. doi:10.1001/archneur.60.8.1119. PMID 12925369.
"Profiles of General Demographic Characteristics, 2000 Census of Population and Housing, United States" (PDF). U.S. Census Bureau. 2001. Archived (PDF) from the original on 19 August 2008. Retrieved 27 August 2008.
Ferri CP, Prince M, Brayne C, Brodaty H, Fratiglioni L, Ganguli M, Hall K, Hasegawa K, Hendrie H, Huang Y, Jorm A, Mathers C, Menezes PR, Rimmer E, Scazufca M (December 2005). "Global prevalence of dementia: a Delphi consensus study". Lancet. 366 (9503): 2112–7. doi:10.1016/S0140-6736(05)67889-0. PMC 2850264. PMID 16360788.
World Health Organization (2006). Neurological Disorders: Public Health Challenges. Switzerland: World Health Organization. pp. 204–07. ISBN 978-92-4-156336-9. Archived from the original on 10 February 2010.
2006 prevalence estimate:
Brookmeyer R, Johnson E, Ziegler-Graham K, Arrighi HM (July 2007). "Forecasting the global burden of Alzheimer's disease". Alzheimer's & Dementia. 3 (3): 186–91. CiteSeerX 10.1.1.729.847. doi:10.1016/j.jalz.2007.04.381. PMID 19595937. Archived from the original on 7 December 2008. Retrieved 18 June 2008.
"World population prospects: the 2006 revision, highlights" (PDF). Working Paper No. ESA/P/WP.202. Population Division, Department of Economic and Social Affairs, United Nations. 2007. Archived from the original (PDF) on 19 August 2008. Retrieved 27 August 2008.
Auguste D.:
Alzheimer A (1907). "über eine eigenartige Erkrankung der Hirnrinde" [About a peculiar disease of the cerebral cortex]. Allgemeine Zeitschrift für Psychiatrie und Psychisch-Gerichtlich Medizin (in German). 64 (1–2): 146–48.
Alzheimer A (1987). Translated by H. Greenson. "About a peculiar disease of the cerebral cortex. By Alois Alzheimer, 1907 (Translated by L. Jarvik and H. Greenson)". Alzheimer Disease and Associated Disorders. 1 (1): 3–8. PMID 3331112.
Ulrike M, Konrad M (2003). Alzheimer: The Life of a Physician and the Career of a Disease. New York: Columbia University Press. p. 270. ISBN 978-0-231-11896-5.
Berrios GE (1990). "Alzheimer's Disease: A Conceptual History". Int. J. Ger. Psychiatry. 5 (6): 355–65. doi:10.1002/gps.930050603.
Kraepelin Emil (17 January 2007). Clinical Psychiatry: A Textbook For Students And Physicians (Reprint). Translated by Diefendorf A. Ross. Kessinger Publishing. p. 568. ISBN 978-1-4325-0833-3.
Katzman R, Terry RD, Bick KL, eds. (1978). Alzheimer's Disease: Senile Dementia and Related Disorders. New York: Raven Press. p. 595. ISBN 978-0-89004-225-0.
Boller F, Forbes MM (June 1998). "History of dementia and dementia in history: an overview". Journal of the Neurological Sciences. 158 (2): 125–33. doi:10.1016/S0022-510X(98)00128-2. PMID 9702682.
Amaducci LA, Rocca WA, Schoenberg BS (November 1986). "Origin of the distinction between Alzheimer's disease and senile dementia: how history can clarify nosology". Neurology. 36 (11): 1497–9. doi:10.1212/wnl.36.11.1497. PMID 3531918.
Allegri RF, Butman J, Arizaga RL, Machnicki G, Serrano C, Taragano FE, Sarasola D, Lon L (August 2007). "Economic impact of dementia in developing countries: an evaluation of costs of Alzheimer-type dementia in Argentina". International Psychogeriatrics. 19 (4): 705–18. doi:10.1017/S1041610206003784. PMID 16870037.
Suh GH, Knapp M, Kang CJ (August 2006). "The economic costs of dementia in Korea, 2002". International Journal of Geriatric Psychiatry. 21 (8): 722–8. doi:10.1002/gps.1552. PMID 16858741.
Wimo A, Jonsson L, Winblad B (2006). "An estimate of the worldwide prevalence and direct costs of dementia in 2003". Dementia and Geriatric Cognitive Disorders. 21 (3): 175–81. doi:10.1159/000090733. PMID 16401889.
Moore MJ, Zhu CW, Clipp EC (July 2001). "Informal costs of dementia care: estimates from the National Longitudinal Caregiver Study". The Journals of Gerontology. Series B, Psychological Sciences and Social Sciences. 56 (4): S219–28. doi:10.1093/geronb/56.4.S219. PMID 11445614.
Jönsson L, Eriksdotter Jönhagen M, Kilander L, Soininen H, Hallikainen M, Waldemar G, et al. (May 2006). "Determinants of costs of care for patients with Alzheimer's disease". International Journal of Geriatric Psychiatry. 21 (5): 449–59. doi:10.1002/gps.1489. PMID 16676288.
"The MetLife study of Alzheimer's disease: The caregiving experience" (PDF). MetLife Mature Market Institute. August 2006. Archived from the original (PDF) on 8 January 2011. Retrieved 5 February 2011.
Schneider J, Murray J, Banerjee S, Mann A (August 1999). "EUROCARE: a cross-national study of co-resident spouse carers for people with Alzheimer's disease: I--Factors associated with carer burden". International Journal of Geriatric Psychiatry. 14 (8): 651–61. doi:10.1002/(SICI)1099-1166(199908)14:8<651::AID-GPS992>3.0.CO;2-B. PMID 10489656.
Murray J, Schneider J, Banerjee S, Mann A (August 1999). "EUROCARE: a cross-national study of co-resident spouse carers for people with Alzheimer's disease: II--A qualitative analysis of the experience of caregiving". International Journal of Geriatric Psychiatry. 14 (8): 662–7. doi:10.1002/(SICI)1099-1166(199908)14:8<662::AID-GPS993>3.0.CO;2-4. PMID 10489657.
Zhu CW, Sano M (2006). "Economic considerations in the management of Alzheimer's disease". Clinical Interventions in Aging. 1 (2): 143–54. doi:10.2147/ciia.2006.1.2.143. PMC 2695165. PMID 18044111.
Gaugler JE, Kane RL, Kane RA, Newcomer R (April 2005). "Early community-based service utilization and its effects on institutionalization in dementia caregiving". The Gerontologist. 45 (2): 177–85. doi:10.1093/geront/45.2.177. PMID 15799982.
Ritchie K, Lovestone S (November 2002). "The dementias". Lancet. 360 (9347): 1759–66. doi:10.1016/S0140-6736(02)11667-9. PMID 12480441.
Brodaty H, Hadzi-Pavlovic D (September 1990). "Psychosocial effects on carers of living with persons with dementia". The Australian and New Zealand Journal of Psychiatry. 24 (3): 351–61. doi:10.3109/00048679009077702. PMID 2241719.
Donaldson C, Tarrier N, Burns A (April 1998). "Determinants of carer stress in Alzheimer's disease". International Journal of Geriatric Psychiatry. 13 (4): 248–56. doi:10.1002/(SICI)1099-1166(199804)13:4<248::AID-GPS770>3.0.CO;2-0. PMID 9646153.
Pusey H, Richards D (May 2001). "A systematic review of the effectiveness of psychosocial interventions for carers of people with dementia". Aging & Mental Health. 5 (2): 107–19. doi:10.1080/13607860120038302. PMID 11511058.
Bayley J (2000). Iris: A Memoir of Iris Murdoch. London: Abacus. ISBN 978-0-349-11215-2. OCLC 41960006.
Sparks N (1996). The notebook. Thorndike, Maine: Thorndike Press. p. 268. ISBN 978-0-7862-0821-0.
"Thanmathra". Webindia123.com. Archived from the original on 6 November 2007. Retrieved 24 January 2008.
Ogiwara H (2004). Ashita no Kioku (in Japanese). Tōkyō: Kōbunsha. ISBN 978-4-334-92446-1. OCLC 57352130.
Munro A (2001). Hateship, Friendship, Courtship, Loveship, Marriage: Stories. New York: A.A. Knopf. ISBN 978-0-375-41300-1. OCLC 46929223.
"Malcolm and Barbara: A love story". Dfgdocs. Archived from the original on 24 May 2008. Retrieved 24 January 2008.
"Malcolm and Barbara: A love story". BBC Cambridgeshire. Archived from the original on 10 November 2012. Retrieved 2 March 2008.
Plunkett J (7 August 2007). "Alzheimer's film-maker to face ITV lawyers". London: Guardian Media. Archived from the original on 15 January 2008. Retrieved 24 January 2008.
Cummings JL, Morstorf T, Zhong K (July 2014). "Alzheimer's disease drug-development pipeline: few candidates, frequent failures". Alzheimer's Research & Therapy. 6 (4): 37. doi:10.1186/alzrt269. PMC 4095696. PMID 25024750.
Gutis, Phillip S. (22 March 2019). "An Alzheimer's Drug Trial Gave Me Hope, and Then It Ended". The New York Times. Retrieved 25 March 2019.
Lashuel HA, Hartley DM, Balakhaneh D, Aggarwal A, Teichberg S, Callaway DJ (November 2002). "New class of inhibitors of amyloid-beta fibril formation. Implications for the mechanism of pathogenesis in Alzheimer's disease". The Journal of Biological Chemistry. 277 (45): 42881–90. doi:10.1074/jbc.M206593200. PMID 12167652.
Dodel R, Neff F, Noelker C, Pul R, Du Y, Bacher M, Oertel W (March 2010). "Intravenous immunoglobulins as a treatment for Alzheimer's disease: rationale and current evidence". Drugs. 70 (5): 513–28. doi:10.2165/11533070-000000000-00000. PMID 20329802. Archived from the original on 17 September 2011.
Vaccination:
Hawkes CA, McLaurin J (November 2007). "Immunotherapy as treatment for Alzheimer's disease". Expert Review of Neurotherapeutics. 7 (11): 1535–48. doi:10.1586/14737175.7.11.1535. PMID 17997702.
Solomon B (June 2007). "Clinical immunologic approaches for the treatment of Alzheimer's disease". Expert Opinion on Investigational Drugs. 16 (6): 819–28. doi:10.1517/13543784.16.6.819. PMID 17501694.
Woodhouse A, Dickson TC, Vickers JC (2007). "Vaccination strategies for Alzheimer's disease: A new hope?". Drugs & Aging. 24 (2): 107–19. doi:10.2165/00002512-200724020-00003. PMID 17313199.
"Study Evaluating ACC-001 in Mild to Moderate Alzheimers Disease Subjects". Clinical Trial. US National Institutes of Health. 11 March 2008. Archived from the original on 30 July 2013. Retrieved 5 June 2008.
"Study Evaluating Safety, Tolerability, and Immunogenicity of ACC-001 in Subjects with Alzheimer's Disease". US National Institutes of Health. Archived from the original on 29 October 2008. Retrieved 5 June 2008.
"Alzheimer's Disease Vaccine Trial Suspended on Safety Concern". Medpage Today. 18 April 2008. Archived from the original on 23 April 2008. Retrieved 14 June 2008.
"Bapineuzumab in Patients with Mild to Moderate Alzheimer's Disease/ Apo_e4 Non-carriers" (Clinical Trial). US National Institutes of Health. 29 February 2008. Archived from the original on 22 March 2008. Retrieved 23 March 2008.
Sperling RA, Jack CR, Black SE, Frosch MP, Greenberg SM, Hyman BT, Scheltens P, Carrillo MC, Thies W, Bednar MM, Black RS, Brashear HR, Grundman M, Siemers ER, Feldman HH, Schindler RJ (July 2011). "Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer's Association Research Roundtable Workgroup". Alzheimer's & Dementia. 7 (4): 367–85. doi:10.1016/j.jalz.2011.05.2351. PMC 3693547. PMID 21784348.
"Safety, Tolerability and Efficacy Study to Evaluate Subjects with Mild Cognitive Impairment" (Clinical Trial). US National Institutes of Health. 11 March 2008. Archived from the original on 22 October 2008. Retrieved 23 March 2008.
"Study Evaluating the Safety, Tolerability and Efficacy of PBT2 in Patients with Early Alzheimer's Disease" (Clinical Trial). US National Institutes of Health. 13 January 2008. Archived from the original on 31 August 2008. Retrieved 23 March 2008.
Etanercept research:
Tobinick E (September 2009). "Tumour necrosis factor modulation for treatment of Alzheimer's disease: rationale and current evidence". CNS Drugs. 23 (9): 713–25. doi:10.2165/11310810-000000000-00000. PMID 19689163.
Tobinick E, Gross H, Weinberger A, Cohen H (April 2006). "TNF-alpha modulation for treatment of Alzheimer's disease: a 6-month pilot study". MedGenMed. 8 (2): 25. PMC 1785182. PMID 16926764.
Griffin WS (January 2008). "Perispinal etanercept: potential as an Alzheimer therapeutic". Journal of Neuroinflammation. 5: 3. doi:10.1186/1742-2094-5-3. PMC 2241592. PMID 18186919.
Tobinick E (December 2007). "Perispinal etanercept for treatment of Alzheimer's disease". Current Alzheimer Research. 4 (5): 550–2. doi:10.2174/156720507783018217. PMID 18220520.
Cheng X, Shen Y, Li R (November 2014). "Targeting TNF: a therapeutic strategy for Alzheimer's disease". Drug Discovery Today. 19 (11): 1822–1827. doi:10.1016/j.drudis.2014.06.029. PMID 24998784.
Wischik CM, Bentham P, Wischik DJ, Seng KM (July 2008). "Tau aggregation inhibitor (TAI) therapy with remberTM arrests disease progression in mild and moderate Alzheimer's disease over 50 weeks". Alzheimer's & Dementia. 4 (4): T167. doi:10.1016/j.jalz.2008.05.438. Retrieved 30 July 2008.
Harrington C, Rickard J, Horsley D (July 2008). "Methylthioninium chloride (MTC) acts as a tau aggregation inhibitor (TAI) in a cellular model and reverses tau pathology in transgenic mouse models of Alzheimer's disease". Alzheimer's & Dementia. 4 (4): T120–T121. doi:10.1016/j.jalz.2008.05.259.
Doody RS, Gavrilova SI, Sano M, Thomas RG, Aisen PS, Bachurin SO, Seely L, Hung D (July 2008). "Effect of dimebon on cognition, activities of daily living, behaviour, and global function in patients with mild-to-moderate Alzheimer's disease: a randomised, double-blind, placebo-controlled study". Lancet. 372 (9634): 207–15. doi:10.1016/S0140-6736(08)61074-0. PMID 18640457.
Bezprozvanny I (October 2010). "The rise and fall of Dimebon". Drug News & Perspectives (Original article). 23 (8): 518–23. doi:10.1358/dnp.2010.23.8.1500435. PMC 3922928. PMID 21031168.
"Pfizer And Medivation announce results from two phase 3 studies in Dimebon (latrepirdine*) Alzheimer's disease clinical development program (NASDAQ:MDVN)" (Press release). Archived from the original on 4 September 2012. Retrieved 16 November 2012.
Wendler A, Wehling M (March 2012). "Translatability scoring in drug development: eight case studies". Journal of Translational Medicine. 10 (10): 39. doi:10.1186/1479-5876-10-39. PMC 3330010. PMID 22397594.
Baddeley TC, McCaffrey J, Storey JM, Cheung JK, Melis V, Horsley D, Harrington CR, Wischik CM (January 2015). "Complex disposition of methylthioninium redox forms determines efficacy in tau aggregation inhibitor therapy for Alzheimer's disease". The Journal of Pharmacology and Experimental Therapeutics. 352 (1): 110–8. doi:10.1124/jpet.114.219352. PMID 25320049.
Wischik CM, Harrington CR, Storey JM (April 2014). "Tau-aggregation inhibitor therapy for Alzheimer's disease". Biochemical Pharmacology. 88 (4): 529–39. doi:10.1016/j.bcp.2013.12.008. PMID 24361915.
Marciniak R, Sheardova K, Cermáková P, Hudeček D, Sumec R, Hort J (2014). "Effect of meditation on cognitive functions in context of aging and neurodegenerative diseases". Frontiers in Behavioral Neuroscience. 8: 17. doi:10.3389/fnbeh.2014.00017. PMC 3903052. PMID 24478663.
Larouche E, Hudon C, Goulet S (January 2015). "Potential benefits of mindfulness-based interventions in mild cognitive impairment and Alzheimer's disease: an interdisciplinary perspective". Behavioural Brain Research. 276 (276): 199–212. doi:10.1016/j.bbr.2014.05.058. PMID 24893317.
Jaunmuktane Z, Mead S, Ellis M, Wadsworth JD, Nicoll AJ, Kenny J, et al. (September 2015). "Evidence for human transmission of amyloid-β pathology and cerebral amyloid angiopathy". Nature. 525 (7568): 247–50. Bibcode:2015Natur.525..247J. doi:10.1038/nature15369. PMID 26354483.
Abbott A (September 2015). "Autopsies reveal signs of Alzheimer's in growth-hormone patients". Nature. 525 (7568): 165–6. Bibcode:2015Natur.525..165A. doi:10.1038/525165a. PMID 26354460.
Martin C, Solís L, Concha MI, Otth C (June 2011). "[Herpes simplex virus type 1 as risk factor associated to Alzheimer disease]" [Herpes Simplex Virus Type 1 as Risk Factor Associated to Alzheimer Disease]. Revista Medica de Chile (in Spanish). 139 (6): 779–86. doi:10.4067/S0034-98872011000600013. PMID 22051760.
Wozniak MA, Mee AP, Itzhaki RF (January 2009). "Herpes simplex virus type 1 DNA is located within Alzheimer's disease amyloid plaques". The Journal of Pathology (Original study). 217 (1): 131–8. doi:10.1002/path.2449. PMID 18973185.
Itzhaki RF (2014). "Herpes simplex virus type 1 and Alzheimer's disease: increasing evidence for a major role of the virus". Frontiers in Aging Neuroscience. 6: 202. doi:10.3389/fnagi.2014.00202. PMC 4128394. PMID 25157230.
Itzhaki RF, Lathe R, Balin BJ, Ball MJ, Bearer EL, Braak H, et al. (2016). "Microbes and Alzheimer's Disease". Journal of Alzheimer's Disease. 51 (4): 979–84. doi:10.3233/JAD-160152. PMC 5457904. PMID 26967229. Archived from the original on 10 November 2016.
Alonso R, Pisa D, Rábano A, Carrasco L (July 2014). "Alzheimer's disease and disseminated mycoses". European Journal of Clinical Microbiology & Infectious Diseases. 33 (7): 1125–32. doi:10.1007/s10096-013-2045-z. PMID 24452965.
Pisa D, Alonso R, Rábano A, Rodal I, Carrasco L (October 2015). "Different Brain Regions are Infected with Fungi in Alzheimer's Disease". Scientific Reports. 5: 15015. Bibcode:2015NatSR...515015P. doi:10.1038/srep15015. PMC 4606562. PMID 26468932.
"Fungus, the bogeyman". The Economist. 22 October 2015. Archived from the original on 8 August 2017.
Kumar DK, Choi SH, Washicosky KJ, Eimer WA, Tucker S, Ghofrani J, Lefkowitz A, McColl G, Goldstein LE, Tanzi RE, Moir RD (May 2016). "Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer's disease". Science Translational Medicine. 8 (340): 340ra72. doi:10.1126/scitranslmed.aaf1059. PMC 5505565. PMID 27225182.
Kolata, Gina (25 May 2016). "Could Alzheimer's Stem From Infections? It Makes Sense, Experts Say". The New York Times. The New York Times. Archived from the original on 4 February 2017.
"Alzheimer's culprit may fight other diseases". Science News. 16 June 2016. Archived from the original on 26 May 2016.
Dougall NJ, Bruggink S, Ebmeier KP (2004). "Systematic review of the diagnostic accuracy of 99mTc-HMPAO-SPECT in dementia". The American Journal of Geriatric Psychiatry. 12 (6): 554–70. doi:10.1176/appi.ajgp.12.6.554. PMID 15545324.
Carpenter AP, Pontecorvo MJ, Hefti FF, Skovronsky DM (August 2009). "The use of the exploratory IND in the evaluation and development of 18F-PET radiopharmaceuticals for amyloid imaging in the brain: a review of one company's experience". The Quarterly Journal of Nuclear Medicine and Molecular Imaging. 53 (4): 387–93. PMID 19834448.
Leung K (8 April 2010). "(E)-4-(2-(6-(2-(2-(2-(18F-fluoroethoxy)ethoxy)ethoxy)pyridin-3-yl)vinyl)-N-methyl benzenamine [[18F]AV-45]". Molecular Imaging and Contrast Agent Database. Archived from the original on 7 June 2010. Retrieved 24 June 2010.
Rabinovici GD, Jagust WJ (2009). "Amyloid imaging in aging and dementia: testing the amyloid hypothesis in vivo". Behavioural Neurology. 21 (1): 117–28. doi:10.3233/BEN-2009-0232. PMC 2804478. PMID 19847050. Archived from the original on 30 July 2013.
O'Brien JT (December 2007). "Role of imaging techniques in the diagnosis of dementia". The British Journal of Radiology. 80 Spec No 2 (Spec No 2): S71–7. doi:10.1259/bjr/33117326. PMID 18445747.
Clark CM, Schneider JA, Bedell BJ, Beach TG, Bilker WB, Mintun MA, et al. (January 2011). "Use of florbetapir-PET for imaging beta-amyloid pathology". JAMA. 305 (3): 275–83. doi:10.1001/jama.2010.2008. PMID 21245183.
"Amyvid". Community register of medicinal products for human use. European Community. 17 January 2013. Retrieved 18 April 2018.
Chong MS, Sahadevan S (September 2005). "Preclinical Alzheimer's disease: diagnosis and prediction of progression". The Lancet. Neurology. 4 (9): 576–9. doi:10.1016/s1474-4422(05)70168-x. PMID 16109364.
Sharma N, Singh AN (July 2016). "Exploring Biomarkers for Alzheimer's Disease". Journal of Clinical and Diagnostic Research (Review). 10 (7): KE01–6. doi:10.7860/JCDR/2016/18828.8166. PMC 5020308. PMID 27630867. |