概要
由于该组织的重要功能和美学作用,皮肤伤口的治疗是一个关键的研究领域。当皮肤受损时,细菌很快就会渗入下层组织,导致危及生命的感染。因此,有效的治疗方法对于处理这种病理状况是必要的。最近,装有抗微生物剂的伤口敷料已成为减少伤口细菌定植和感染的可行选择,以改善愈合过程。在本文中,我们概述了最突出的抗生素嵌入式伤口敷料,以及它们的使用限制。可以整合到伤口敷料中的一种有希望但仍被低估的潜在抗菌剂组是天然产品,尤其是精油。本文介绍了一些最常用的抗多种药物的精油,如茶树,圣约翰草,薰衣草和牛至,以及它们掺入伤口敷料中。此外,另一种具有令人鼓舞的抗菌活性的天然产品是蜂蜜。我们重点介绍了世界不同地区的研究人员对浸渍蜂蜜的伤口敷料进行的几项研究的最新结果,特别强调了麦卢卡蜂蜜。最后,我们重点介绍了使用纳米粒子作为平台的最新进展,以增加针对伤口愈合的药物制剂的效果。单独的银,金和锌纳米颗粒或用各种抗微生物化合物功能化已经整合到伤口敷料中并且证明了对伤口的治疗效果。
关键词:纳米粒,伤口愈合,伤口感染,抗生素,抗菌活性精油,麦卢卡蜂蜜
1.简介
伤口可以代表器官(例如皮肤)或组织的简单或严重的病症,并且可以扩散到其他组织和解剖结构(例如,皮下组织,肌肉,肌腱,神经,血管,甚至骨骼 )[1]。在所有人体(HB)器官中,皮肤无疑是最容易受到损伤和伤害,划痕和烧伤的。通过破坏上皮和结缔组织,HB的外部环境保护能力被削弱。因此,必须重新制作功能性表皮或甚至其他皮肤层。这发生在一系列交叉阶段,称为伤口愈合或伤口修复。 HB的能力可以用可行的皮肤结构替代丢失的皮肤结构,并形成疤痕。
不正确的修复过程会造成严重的损害,如皮肤的损失,感染的发生,以及对下层组织甚至是全身组织的伤害[2]。伤口愈合的最常见和不可避免的障碍是感染的安装,主要是在慢性伤口的情况下。虽然细菌是完整皮肤微生物群和伤口的常见部分,但现有细菌的临界阈值和生物膜的形成可能会阻碍伤口愈合[3]。由于这些事实,无论最近在伤口处理方面取得了哪些进展,细菌和真菌感染仍被认为是导致显着死亡率和发病率的最集体和最痛苦的状态之一[4]。金黄色葡萄球菌,耐甲氧西林金黄色葡萄球菌(MRSA)和铜绿假单胞菌是在感染伤口的患者中发生的主要微生物菌株[5]。
由于独特的生物,非无菌伤口环境和极其复杂的伤口愈合系统,仍然需要有效和有针对性的治疗。因此,目前的研究动机是为慢性和急性伤口感染寻找更有效的治疗方法[6]。在慢性伤口发生的情况下,个体经常忍受广泛的治疗和定期换药,完全可溶解的,不可替换的或非粘附的伤口敷料,以精确的方式将治疗分布到伤口部位可以改善治疗结果和伤口对药物的反应[7]。
伤口敷料通常仅用于保护伤口免受外部污染,但它们也可以用不同的治疗复合物功能化以递送到伤口部位[8]。与在愈合过程中没有活性功能的传统敷料(如棉和羊毛制成的绷带)相比,先进的伤口敷料可以通过掺入的活性成分[9]设计成参与其中。整合的复合物必须在伤口愈合过程中显示出动态作用,或者有助于去除坏死组织,预防/治疗已安装的感染,或两者兼而有之。在这方面,敷料可以通过多种抗生素(如喹诺酮类[10],四环素类[11],氨基糖苷类[12],头孢菌素类[13]等)或其他具有抗菌特性的物质(如必需品)进行功能化)[14,15]。抗生素嵌入式伤口敷料对局部感染的治疗很有价值,因为局部需要高浓度的抗生素[16]。
但是,在某些情况下,大量抗生素会导致全身毒性[17]。新抗生素的开发在过去几年中有所减少,少数公司在这些领域仍然活跃。此外,由于抗生素的过度使用和误用,抗生素抗性微生物大大增加[18]。这种抗生素危机仍在进行中,并影响用于全身和局部感染的抗生素治疗。例如,慢性伤口(在糖尿病足,静脉溃疡和压力性溃疡的情况下)的治疗通常涉及长期治疗。鉴于这些感染引起的当前问题,非常规非抗生素治疗的抗微生物潜力具有新的兴趣。特别是,天然药物,特别是精油[19,20]和蜂蜜在伤口愈合中的作用吸引了科学界以及一些生产伤口敷料的公司[21]。
此外,纳米技术可以成为规避高全身剂量的真正支持。纳米技术代表了一个发展领域,操纵纳米尺寸范围或分子/原子尺度的材料,这些材料可以在再生医学和预防各种疾病中有许多应用[22]。当将材料尺寸减小到纳米尺度时,其表面与面积/体积比将急剧增加,从而为无数医疗应用产生有利的物理化学反应性。医学用途的一些实例包括新药物的开发和更多的手术药物递送平台,体内成像,纳米生物传感器,伤口愈合等[23]。在纳米医学中,金属纳米颗粒(NPs)如银(Ag)[24],金(Au)[25]和锌(Zn)[26]显示出令人印象深刻的特性,如低体内毒性和抑菌/杀菌活性[27]。 。这些方便的特性推荐将它们整合到伤口敷料中。
本文旨在回顾在包含抗生素,精油(EO)和预防和/或治疗感染伤口的蜂蜜等活性成分的伤口敷料领域中值得注意的体外和体内结果。与此同时,我们收集并纳入了最近的数据,这些数据强调了基于NPs的伤口敷料的无与伦比的潜力,这些伤口敷料可以优化伤口敷料的治疗效果。
2.伤口愈合过程和皮肤伤口微生物学
皮肤是HB中最大的器官,是防御机械,化学和生物制剂以及紫外线辐射的防御屏障。 此外,它会出现过多的水分流失,提供水合作用和温度调节[28]。 皮肤有三个多组织层,即表皮,真皮和皮下组织,以及皮肤辅助(如毛和腺),从深层真皮生长到浅层表皮层[29]。
任何组织中的伤口愈合都是正常的生物过程,它涉及四个复杂的步骤:体内平衡/凝固; 炎症,迁移和增殖; 再上皮化和恢复[30](图1)。
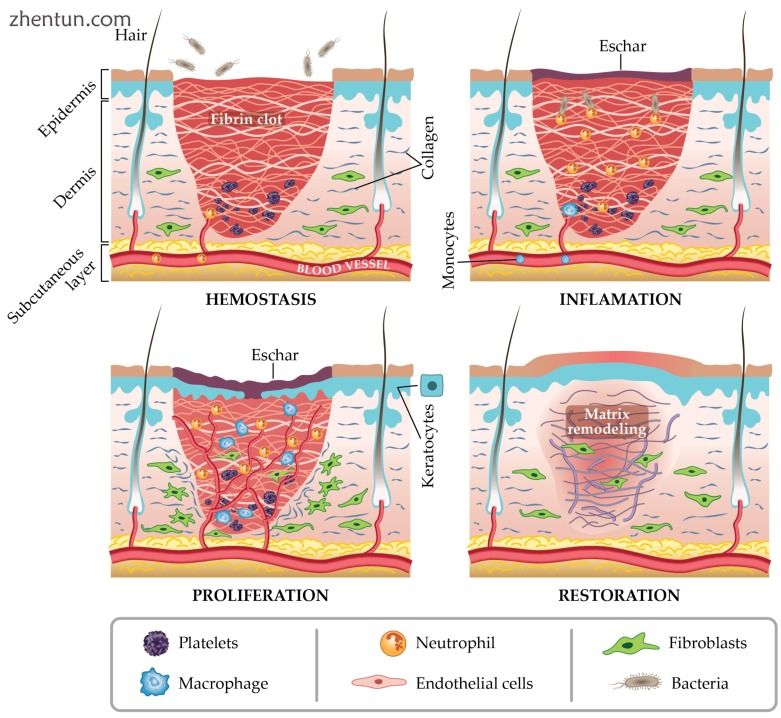
图1
伤口修复的四个阶段。
伤口愈合过程的每个阶段都受到一系列基本介质的影响,如血小板和细胞因子,炎症细胞,细胞和细胞外基质,蛋白酶,生长因子和抑制剂[31]。通常,止血和炎症阶段在损伤后立即发生,但炎症阶段可持续长达6天[2]。增殖期被认为是血管生成和细胞外基质发育的开始[32]。炎症和/或增殖期的延长时间将导致阻碍愈合,促进过度瘢痕组织的建立[33]。重塑阶段通常在损伤后3周开始,并且可能需要长达2年才能完全解决[34]。关于伤口愈合复杂过程的许多细节可以在Martin和Nunan最近的报告中找到[35]。对这一阶段序列的综合考虑,每个阶段涉及的细胞,时间表和分子信号传导,可以同意最大程度地优化愈合治疗。
伤口可分为急性和慢性。急性伤口代表受伤的皮肤(例如,由烧伤和化学伤引起的),其通过伤口修复的常规阶段愈合;相反,慢性伤口需要更长的愈合时间。这种较长的愈合时间通常可归因于许多因素,包括炎症介质的放大水平,伤口感染,缺氧和营养不良[36]。同时,它可能取决于患者的年龄[37]或潜在的合并症(如糖尿病,伤口干燥)[38]。
皮肤微生物群的多样性和皮肤微环境(干燥,潮湿和皮脂)可以影响伤口修复过程和皮肤感染的发生[39]。皮肤上存在四种主要的细菌门:放线菌,变形菌,厚壁菌和拟杆菌,它们排列成生物膜并积极参与抑制皮肤感染[40]。
一旦皮肤受损,正常皮肤菌群和外源细菌和真菌的典型微生物很快就会进入下层组织,这为其发育提供了一个潮湿,温暖和营养丰富的环境[41]。 然而,当愈合延迟时,伤口的正常微生物群会发生变化,并且会产生更具攻击性的微生物类型[42]。 因此,开放性伤口可以是微生物增殖和定植的有利位置。 在慢性伤口构建的初始阶段,革兰氏阳性实体(主要是金黄色葡萄球菌)出现最多。 在晚期阶段,革兰氏阴性物种(例如,大肠杆菌和假单胞菌属物种)大多存在并且可能进入更深的皮肤层,显着影响组织。 此外,约50%的慢性伤口存在球菌类型[5]。 伤口部位的感染始于污染,并继续(急性)定植和伤口感染(如图2所示)。
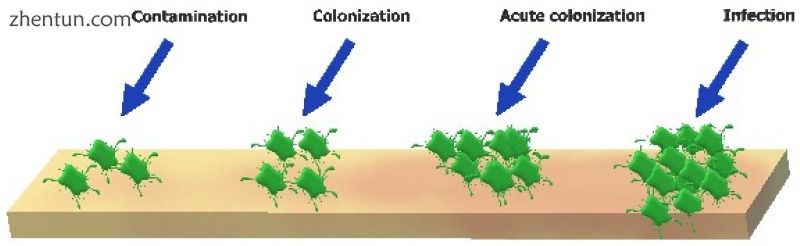
图2
从污染到伤口感染的连续体的插图。污染以非复制性细菌的存在为代表,是慢性伤口的常见情况;伤口污染不会促进愈合受损。定殖是复制微生物的发生而不引发宿主反应。急性定植的特征在于复制细菌的表现,导致适度的局部反应;生物负载的这种增加可以延缓伤口愈合。当微生物繁殖并进入组织时发生感染,产生全身宿主反应。
伤口的污染和定植都不会引发免疫反应。除了微生物细胞的增殖外,急性定植通常与扩大的局部疼痛和炎症反应的开始有关[43]。通过微生物侵入健康组织,产生了一系列局部和全身宿主反应,例如化脓性排出,扩散性红斑或症状性蜂窝织炎[3]。
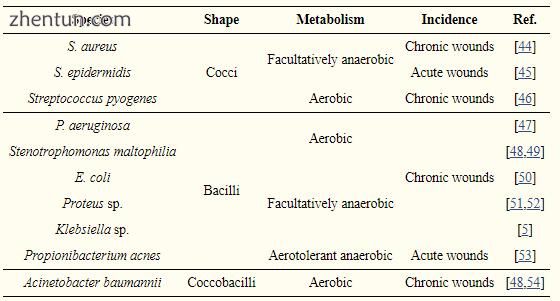
如上所述,由于加​​重和延长的炎症阶段,微生物的无限制发展可延迟伤口闭合。有问题的病原体总结在表1中。
表格1
在急性和慢性伤口中形成的微生物种类。
慢性伤口中的感染通常是多微生物的,促进协同效应(需氧和厌氧微生物维持彼此的持久性和增殖)。这种集体效应通常可以通过氧气消耗来支持;好氧细菌可以促进组织缺氧,为厌氧增殖创造有利条件。一旦建立了厌氧物种,它们就可以通过产生短链脂肪酸来阻止其他微生物的吞噬作用。此外,来自一种细菌的营养物质通量可以维持另一种细菌的进化和增殖[3]。在许多类型的慢性伤口中,金黄色葡萄球菌和铜绿假单胞菌通常在共培养中同时生长[42]。许多病原体可以粘在一起,形成生物膜,生物膜是被聚合物环境包围的微生物块,因此逃避了抗生素和宿主效应物的杀灭活性。生物膜可以被认为是伤口愈合的物理障碍,其中炎性期的常规过程可以延长。据报道,脂肪酸形式的细菌副产物可以阻止中性粒细胞的趋化性和大肠杆菌和金黄色葡萄球菌细菌细胞的吞噬作用[55,56]。 MRSA感染使身体易受其他形式的细菌和真菌感染,并且及时导致多物种伤口感染的产生,这些感染有待消除[57]。例如,MRSA代表40%的伤口分离物,感染14-17%的烧伤患者[58]。此外,发现在许多情况下,念珠菌属。共同感染烧伤[59]。
在免疫受损的个体中,细菌可以进入更深的组织。为了加速伤口愈合,局部抗菌药物可以促进严重感染伤口的治疗。体外试验和从患者收集的数据表明,抗菌伤口敷料可能有助于可能受生物膜影响的伤口[60,61]。
虽然抗生素通过治疗感染显着改善了人类的健康,但许多上述感染菌株仍然通过抗菌素耐药性在世界范围内(在医院和社区环境中)引起严重问题[4]。随着抗微生物药物耐药性的增加,天然产物,改性抗生素,纳米颗粒等形式的新型抗微生物剂可以对抗微生物抗性病原体的试验产生积极反应。
3.伤口敷料的理想特性
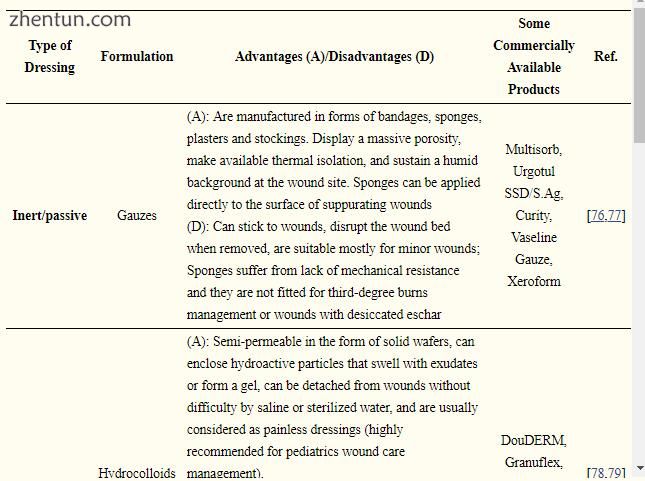
为了解决与伤口感染相关的健康问题,已经开发了不同形式的伤口敷料以保护伤口免受污染并且还加速伤口愈合。最近,被动地提供伤口安全性的传统伤口敷料(例如棉和羊毛的绷带)已经被创新的敷料所取代,这些敷料精通提供有利的环境并将有效成分分配到伤口以促进伤口愈合。在这方面,已经使用了大量的合成或天然材料和组合,具有多种配方(例如,海绵[10],水凝胶[62],薄膜[63,64,65],水胶体[66],水纤维垫[67,68]等)和使其适合特定伤口处理的特性。根据其作用性质,伤口敷料分为三大类:惰性/被动,生物活性和交互性(表2)。
表2
伤口敷料类型。
当与伤口接触时,敷料必须提供潮湿的环境,同时吸收伤口液[69,70,71],并保持适当的组织温度以改善血液流向伤口[72]。 敷料必须是生物相容的[73],对水和氧气是半透性的[74],促进组织更新过程,低过敏性而不引起免疫反应[72](图3)。 此外,敷料在移除时不得产生创伤,并且还必须具有成本效益。 可以得出结论,由于每个伤口的不同特征,包括愈合阶段,在所有情况下都没有适合的敷料。 然而,许多研究团队制造和改进了不同的伤口敷料材料,以满足特定伤口阶段的大部分需求[75]。
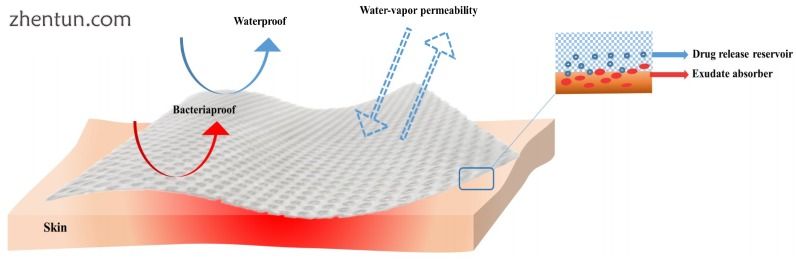
图3
理想伤口敷料的性质。
最近的进展致力于将抗微生物/抗菌剂添加到传统的伤口敷料本身中。含有抗菌剂的伤口敷料不建议用于去除扩散感染(通常涉及全身性抗生素治疗),或用于治疗未感染的伤口床。尽管如此,它们大多适合局部感染的伤口[85]。
4.伤口敷料中的抗菌剂
4.1.抗生素
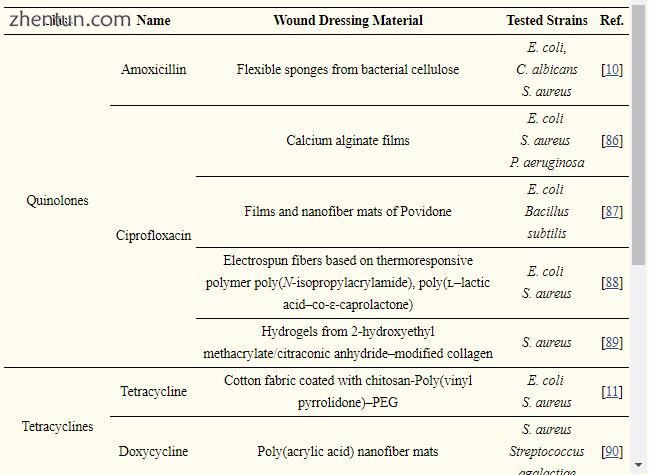
许多研究表明,各种抑菌或杀菌抗生素可以帮助伤口闭合,但它们对伤口愈合的积极影响仍然经常被忽视。即使已知无数抗生素对产生感染的微生物有效,但仅使用喹诺酮类,四环素类,氨基糖苷类和头孢菌素类来生产抗菌伤口敷料。表3总结了含抗生素的伤口敷料及其作用范围。
表3
伤口敷料中含有抗生素。
这些类别的抗生素可以通过以下四种途径之一阻碍细菌的某些功能和/或代谢途径:抑制细菌细胞壁合成,阻断关键代谢途径,干扰蛋白质合成和抑制核酸合成[ 93,94]。尽管许多抗生素可用于治疗感染的伤口,但它们的重复和/或不当使用可引发细菌耐药[95]。结果发现,大约70%的导致伤口感染的细菌对最常用的一种抗生素有抗药性[96]。根据对470个具有细菌学鉴定的伤口分泌物样本进行的研究,金黄色葡萄球菌和铜绿假单胞菌菌株对抗生素应用具有显着的弹性[97]。此外,传染性菌株开始对几乎所有类别的抗生素产生抗性,导致迫切需要寻找新的治疗替代品,如EO [98]和NPs [95]。
4.2.用于伤口感染的天然抗菌药物
随着抗生素逐渐被产生感染的菌株所容忍,人们现在正在吸引大量的生物资源。这些主要是草药,但可能包括动物和矿物成分。文献报道了许多对多种微生物感染并发创伤的天然药物[99]。它们的杀菌效果(在生物膜水平)针对伤口感染的初始阶段和晚期阶段[100]。
本节包含最近发表的文献,其中涉及EO和其他天然替代品(如蜂蜜)的应用,作为促进伤口愈合过程的再生和抗菌剂。许多体外和体内研究证实了必需和/或食用油和蜂蜜的抗微生物和再生特性。
4.2.1.精油
EO,也称为“挥发性天然混合物”,是植物次生代谢产物,具有抗氧化,抗炎,抗过敏,抗病毒,抗菌和再生的特性[101]。 EO主要由植物的植物部分(例如叶子,种子,树皮,树枝和根)合成。
各种研究指出,EO在伤口敷料中的抗菌作用可归因于它们的不同成分(如肉桂醛,香叶醇,百里香酚,薄荷醇和香芹酚等)[21,102,103]。 EO中这些成分的含量和存在高度依赖于萃取程序(例如,水蒸馏,微波辅助萃取,蒸汽蒸馏,微波产生的水蒸气蒸馏,微波蒸汽扩散和超声波辅助萃取)[19]和样品来源[20]。关于EO对酚类化合物(特别是百里香酚和香芹酚)的抗菌活性的多样化发表论文详细解释了这些化合物对细菌起作用的机制。例如,Kavoosi等人。 [104],EOs表示EOs攻击存在于细胞膜和细菌细胞壁中的脂质和磷脂,导致细胞质流出,pH降低和细胞过程受损(如ATP生物合成,DNA转录和蛋白质合成)。其他研究小组描述了EOs通过干扰营养物质通过细胞膜的动态转运以及细菌细胞物质的凝固来破坏细胞质膜的目的[105]。
与抗生素相比,EO对抗菌药物耐药性的发展没有/影响很小[106]。此外,有大量的体外试验指出使用EO作为多药物弹性微生物的创新疗法[107,108]。例如,已发现百里香,薄荷,薰衣草,肉桂,茶树,迷迭香,桉树,柠檬草等的EOs具有抗菌特性[109]。除抗生素和防腐剂外,EO还被整合到伤口敷料中,起抗菌作用[110,111]。然而,尽管它们有助于治疗伤口感染,但可能需要重复施用和/或高浓度,这可能导致对患者的不利影响。
互叶白千层(Melaleuca alternifolia),俗称茶树,是民间和传统疗法中的知名植物,由于其作为愈合剂的长期​​历史地位,在现代医学中仍然特别受关注。现在,茶树EO及其组分的有用性质已经可替代地整合到各种产品中,例如皮肤病霜和软膏。从互叶白千层的叶子和末端分枝中提取的茶树EO由约100种不同成分的混合物组成,大多数是单萜和倍半萜[112],其中松油烯-4-醇和1,8-桉树脑是最活跃(镇痛,抗病毒,抗菌,抗真菌,抗原虫,抗炎)。
对于将茶树EO整合到各种功能性敷料中的伤口感染的治疗有令人鼓舞的数据。例如,制备了覆盖有一层壳聚糖并含有茶树EO的电纺聚己内酯(PCL)非织造垫,并在体外进行了研究[113];试验证明,垫子有效地抑制了金黄色葡萄球菌的生长。在未感染和感染的动物模型中,通过在C57BL6 / J小鼠背部具有(1.2×1.2)cm 2尺寸的皮肤扣除进行体内试验。处理后7天,与对照相比,10μL浓度的茶树EO处理组的非织造垫的细菌水平较低:与细菌含量相比,细菌含量减少81.6±18.1%和93.7±9.57%。对照组[113]。其他研究合成了装载有不同含量的茶树EO液滴的壳聚糖膜。所获得的膜在体外针对常见的伤口致病细菌和真菌,金黄色葡萄球菌,大肠杆菌和白色念珠菌进行测试。本研究报道,薄膜对所有菌株都具有抗菌活性,这种活性随着薄膜中茶树EO含量的增加而增强。此外,薄膜对L929成纤维细胞无毒并维持其生长[114]。
在对MRSA菌株并发的慢性​​人类伤口进行随机对照试验,评价茶树EO。在一项不受控制的,开放标签的试验研究中,用于测试茶树EO的有效性,Edmondson等。 [115]招募了19名患有MRSA感染伤口的患者。从19名患者中,11名患者用可与水混溶的茶树油(3.3%)溶液治疗,该溶液在每次换药时施用。尽管治疗后11个伤口中有8个减小了,但感染并未完全消除。在最近的一项研究中,Lee等人。 [116]在养老院居民中使用10%的局部茶树制剂用于MRSA定植的慢性伤口。用茶树EO治疗的16名患者中有14名发生感染,在治疗4周后完全根除。另外,关于伤口愈合过程,与对照组相比,茶树油组中的16个定植伤口在28天内关闭。此外,茶树EO用于生产市售的水凝胶敷料,即Burnaid(Mundipharma Pty Limited,Sydney,Australia),用于治疗烧伤[117]。
贯叶连翘(Stic's Wort)自古以来就被用于对抗抑郁,焦虑,炎症,微生物和疼痛[118]。在过去的几十年中,该植物已被研究其成分,如金丝桃素,hyperforin和类黄酮。贯叶连翘的抗菌活性归因于hyperforin。此外,发现相同的hyperforin在皮肤再上皮化中起主要作用[118]。因此,贯叶连翘的EO已成为伤口治疗中最有效和广泛传播的草药之一[118]。将不同浓度(0.25-1.5%v / v)的贯叶连翘油掺入壳聚糖膜中用于伤口敷料应用。对大肠杆菌和金黄色葡萄球菌菌株进行的体外试验表明,壳聚糖 - 贯叶连翘油基薄膜对两种微生物均具有抗菌作用[119]。
Lavandula angustifolia(俗称薰衣草)已被世界各地的传统医学中使用。已经证明,薰衣草的EO有效抑制了感染微生物的生长[120]。薰衣草EO(LO)的抗菌活性归因于其主要成分芳樟醇和芳樟醇,但薰衣草EO的化学成分和抗菌作用主要取决于薰衣草样品来源[121]。在这方面,Imane等人提出了一项有趣的研究。 [122]谁报道了薰衣草EO的化学成分和抗菌作用。他们的EO证实存在大量芳樟醇,乙酸芳樟酯,1,8-桉叶素和樟脑。此外,体外试验证明了对大肠杆菌ATCC 25922,金黄色葡萄球菌ATCC 25923和铜绿假单胞菌ATCC 27853的强抗菌活性和抑制作用,最小抑制浓度为3.33,1.33和42.67μL/ mL [122]。然而,薰衣草EO被建议用于以预防性或局部应用的形式治疗表面感染[121]。
除了显示的抗菌和抗真菌活性外,薰衣草EO还在改善伤口愈合阶段中起重要作用。值得注意的是提到Mori等人的研究。局部应用LO对受伤大鼠模型的影响,其中LO通过刺激胶原蛋白的产生和成纤维细胞的分化以及通过加速肉芽组织的建立来促进初级阶段的伤口愈合[123]。
牛至属(俗称牛至)有约39种,其中牛至属(Origanum vulgare L.)是许多科学研究中最受关注的[124]。据报道,牛至EO具有多种生物活性,如抗氧化,抗菌和抗突变,与其组成中百里酚和香芹酚的存在相关[124]。许多体外和体内研究表明,EO可用于治疗金黄色葡萄球菌,MRSA和大肠杆菌细菌感染[125]。在Liakos等人的一项工作中,醋酸纤维素电纺纤维有效地掺入了牛至EO,用作改进的抗菌伤口敷料。此外,在分析细菌和生物膜的体外发育过程中,作者在所有研究的EO浓度(1%和5%)中观察到对所有测试菌株的抑制作用,但当5%牛至EO的量为时,这种效果是显着的。用于醋酸纤维素纤维的设计。在用牛至EO形成的纤维上进行的测试显示出对所有测试的微生物菌株具有非常好的抗菌活性,其效力降低如下:白色念珠菌>大肠杆菌>金黄色葡萄球菌[126]。
4.2.2.蜜糖
蜂蜜几十年来一直被用作许多人类疾病的天然治疗剂,例如心血管和胃肠道疾病,上呼吸道感染以及感染的伤口[127]。蜂蜜的治疗特性,如为伤口提供局部营养的能力,炎症减少,肉芽和血管生成刺激,伤口上皮化,建议将其整合到伤口敷料中[21]。在过去几十年中进行的研究表明,蜂蜜的抑菌和杀菌活性可归因于以下几个因素:
-
它是酸性pH(通常在3.4-6.1范围内)。已经发现,蜂蜜的酸性特征可能会促使巨噬细胞根除细菌并抑制微生物生物膜的形成[128]。
-
由糖在其化学成分中发现的渗透压。高渗透压阻碍微生物的发育[129]。
-
存在抗菌成分,如过氧化氢,抗氧化剂,溶菌酶,酚酸,黄酮类,甲基乙二醛和蜂肽(如防御素-1)[130,131]。过氧化氢的产生是抑制细菌发育的关键组分。特别是,当伤口渗出物与葡萄糖氧化相互作用时,过氧化氢逐渐释放/形成,引发对病原体大分子的氧化损伤;过氧化氢可以与细菌细胞壁反应,也可以与细胞内脂质,蛋白质和核酸反应[132]。
考虑到上述情况,对不同蜂蜜类型的研究表明它们对涉及伤口感染的最普遍的微生物具有高效率。 Kuś及其同事[133]发表的调查显示,来自波兰的14种蜂蜜品种的抗菌活性进行了分析,矢车菊,荞麦和百里香蜂蜜对浓度为3.12的金黄色葡萄球菌PCM 2051菌株的生长最为活跃。 6.25%(v / v)。在同一项研究中,观察到对椴树,石南花,咸味和芫荽蜂蜜采取适当的行动。同样,通过使用来自许多地理区域的Ulmo树[134],melaleuca [135]和龙眼花[136]蜂蜜,已经报道了广泛的MRSA微生物的生长抑制。
然而,在存在过氧化氢酶的情况下,降低过氧化氢 - 蜂蜜的酶显示出减少的抗微生物作用。此外,蜂蜜的成分取决于花卉来源,蜜蜂种类和地理环境[137]。为了超越这一限制,以及“传统蜂蜜”产生的问题(例如导致葡萄糖氧化酶失活的孢子的存在),建议仅将具有认证活性的蜂蜜应用于医学领域。其中,各种各样的医用蜂蜜进入市场(例如板栗,麦卢卡,百里香,revamil)[138],与“传统蜂蜜”相比,它具有良好的可预测性和质量。目前,一些公司专注于生产含有蜂蜜(主要是麦卢卡蜂蜜)的敷料:Actilite®,Algivon®,MediHoney®和ActivonTulle®[21]。
麦卢卡蜂蜜是从麦卢卡树中获得的,含有一种非过氧化物,通过过氧化氢酶成分的作用不可降解,可以维持其在生物体液中的抗菌活性[21]。麦卢卡蜂蜜所产生的抗菌特性不仅归因于过氧化氢,还归因于其化学成分中存在的大量抗菌化合物甲基乙二醛[137]。发现麦卢卡蜂蜜阻碍了MRSA和化脓性链球菌的生长,以及测试的革兰氏阴性菌株(如大肠杆菌和铜绿假单胞菌)[139],并且在伤口部位无法建立生物膜[140]。考虑到这种特定类型蜂蜜的有益多方面特性,在抗炎活性,伤口修复功效和抗菌性能方面,许多研究小组制造了麦卢卡蜂蜜敷料。 Minden-Birkenmaier等。他们的研究重点是制造和表征含麦卢卡蜂蜜的聚(ε-己内酯)(PCL)纳米纤维支架,作为伤口敷料和组织工程皮肤的前体。获得的结果证实,蜂蜜对体外成纤维细胞浸润到支架中具有积极影响,同时抑制大肠杆菌菌株的生长[141]。此外,杨和他的同事,将麦卢卡蜂蜜加入到用丝素蛋白生产的静电纺丝膜中作为功能性抗菌剂。获得的纤维基质对MRSA和甲氧西林敏感的金黄色葡萄球菌,大肠杆菌和铜绿假单胞菌具有抗菌活性[142]。在Tavakoli等人的一项研究中,生产出高浓度蜂蜜 - 聚乙烯醇混合水凝胶,不仅可以促进抗菌活性,还可以证明其生物相容性。据观察,蜂蜜/聚(乙烯醇)杂化物通过建立薄层亲水凝胶有效地促进了伤口愈合过程,降低了污染的风险。同样的敷料被证明对金黄色葡萄球菌和大肠杆菌病原体有效[143]。
关于蜂蜜的临床益处,最近的综述概述了观察报告和随机对照试验,以及最近发表的文献[144]的更新。然而,文献报道根据伤口类型改变了蜂蜜的结果,并且已经表明蜂蜜甚至可能产生有害影响。这些不协调的结果表明需要更多的信息,大型随机临床试验对于证明蜂蜜在减少伤口感染流行的临床益处至关重要。
4.3.纳米粒子
治疗由多重耐药细菌产生的伤口感染是一项主要任务,因为常规抗生素不能治疗这种感染[145]。 NP被认为是习惯性应用抗生素的有希望的替代品,因为它们单独表现出对大量病原体的杀菌活性,能够减少药物的副作用并且不产生微生物抗性[27]。
当NP遇到细菌细胞壁时,它可以通过释放有毒金属离子或通过产生活性氧物质(ROS)来实现其杀菌效果[146]。当与细菌细胞壁紧密接触时,源自细菌表面的带负电的基团吸引带正电的NP。那时,建立了范德华力,受体 - 配体和疏水相互作用,并通过在细菌表面建立“孔”来改变细胞壁吸收率[147,148]。此外,NP还可以穿透细胞壁,影响代谢途径,破坏线粒体,并且还可以影响随后的pH修饰和膜表面电荷紊乱的质子外排泵[146,149]。细菌物种的敏感性不仅与其细胞壁的结构有关。额外的因素可以影响细菌对NPs治疗的弱点或耐受性。 NP毒性的机制非常复杂,但通常由NP的组成,表面改性,内在性质和细菌种类决定。例如,大肠杆菌非常容易受到CuO和ZnO NP的影响,而金黄色葡萄球菌和枯草芽孢杆菌则不易受到影响[150]。 Ag NPs对大肠杆菌和金黄色葡萄球菌细菌菌株的抗菌作用高于Cu NPs对抗同种细菌[151,152]。 Cu NPs的毒性受若干因素(例如,高温,高通气,低pH,NP和细菌浓度)的组合控制,这些因素可增加毒性[153]。在用于抗大肠杆菌,枯草芽孢杆菌和金黄色葡萄球菌的CuO和ZnO NP中,CuO NPs具有最高的毒性[96,146]。
许多上述NP单独或用多种抗微生物化合物(例如抗生素和天然产物)功能化已经用于伤口愈合的应用。 Ag NPs受到科学界的广泛关注,因为它们对~650种微生物和抗生素抗性细菌具有抑制作用[24]。 Ag NPs可通过使细菌DNA变性来抑制细菌繁殖,导致细菌细胞改变,最终导致细胞死亡[154]。 Ag和Ag纳米颗粒的毒性可以根除微生物;同样,它可以对健康的人体细胞产生相同的效果。已经发现Ag对人真皮成纤维细胞具有浓度依赖性细胞毒作用[155]。随着纳米技术的扩展,专家们能够建立一个治疗窗口,增强Ag的抗菌性能,降低其最低抑菌浓度,并降低对正常人体细胞的毒性[156]。因此,许多含有Ag的伤口敷料(例如Acticoat,Bactigrass,Tegaderm,Fucidin,PolyMem Silver)已被美国食品和药物管理局[157]接受在市场上的推出。在金属NPs中,Ag NPs广泛应用于配制用于压疮的烧伤和伤口敷料的软膏[158,159]。在使用人类角质形成细胞和真皮成纤维细胞的最新研究中,Ag NPs的作用大大降低了炎性细胞因子的水平并促进了愈合[160]。
现在大量的研究集中在由壳聚糖水凝胶制成的伤口敷料的配方中,所述壳聚糖水凝胶包含并释放纳米Ag和Ag NP [161,162,163,164,165]。例如,将(10-30)nm的球形Ag NP嵌入纳米纤维的表面中以用于伤口愈合应用。该纳米结构显示出显着的抗菌活性,大肠杆菌,金黄色葡萄球菌和铜绿假单胞菌减少超过99%,并且认可表皮细胞的进展而没有细胞毒性[166]。我们小组使用Ag NPs涂覆聚酯 - 尼龙伤口敷料,以降低由铜绿假单胞菌和金黄色葡萄球菌细菌菌株引起的外源性伤口相关感染的风险。体外和体内研究证明,作为包衣应用的测试NP允许培养的成纤维细胞的正常发育并且在小鼠模型内显示出毒性。改良和活细胞计数分析证明,改良的伤口敷料对细菌定植和生物膜生长具有增强的抑制活性,尤其是对铜绿假单胞菌的抑制活性[167]。
由于其复杂的抗菌机制,锌具有抗一些抗生素抗性微生物的活性[168,169]。氧化锌(ZnO)代表了其抗菌作用的核心形式。 ZnO纳米颗粒能够产生阻碍大肠杆菌和金黄色葡萄球菌生物膜发育的ROS [170,171,172]。此外,据报道,适当浓度的ZnO NPs具有抗菌活性而不影响正常细胞[26,173]。当ZnO被嵌入壳聚糖 - 葡聚糖水凝胶中时,它被证明具有适当的抗菌性能并且是伤口敷料中的合适成分[174]。 Knorasani等人。通过将ZnO纳米颗粒掺入肝素化PVA /壳聚糖/水凝胶中制备纳米复合材料敷料。结果表明,与没有ZnO纳米粒子的样品相比,通过在水凝胶中加入ZnO纳米粒子,这种复合敷料对大肠杆菌和金黄色葡萄球菌的抗菌效果提高了70%以上。此外,对小鼠成纤维细胞进行的体外试验表明,纳米复合材料具有完全的生物相容性[175]。
我们的团队创建了一种新型纳米改性涂层,用于伤口敷料,在功能化的ZnO纳米结构和橙油上构建。测试获得的纳米复合材料对大肠杆菌和金黄色葡萄球菌的抗菌效率。结果显示获得的纳米结构对浮游和粘附细菌的有效性,但对金黄色葡萄球菌菌株的作用更强烈[176]。在另一项研究[177]中,蓖麻油被用作基质材料,用于开发填充有不同量的壳聚糖改性的ZnO纳米颗粒的纳米复合膜。针对大肠杆菌,金黄色葡萄球菌和藤黄微球菌评估显影膜的杀微生物性质。对最后两个菌株的抗菌作用强于对大肠杆菌的抗菌作用。此外,细胞活力记录显示壳聚糖-ZnO载量≤5.0wt%的复合物不会对测试的人真皮成纤维细胞产生毒性作用。体内愈合研究表明,用蓖麻油/壳聚糖-ZnO处理的伤口比蓖麻油和纱布覆盖的伤口早愈合,并且表现出更高的含量和更好的排列胶原排列。
众所周知,Au NP具有生物相容性,广泛用于生物医学领域[25,178,179,180]。仅Au的纳米形式不具有抗微生物活性,必须与其他化学物质结合才能有效地应用于抗菌应用[181]。例如,AuNP可以与明胶,壳聚糖和/或胶原蛋白连接以用于伤口愈合[182,183,184]。在一项研究[185]中,将Au NP与冷冻保存的人成纤维细胞结合,局部应用于烧伤;治疗后的伤口表现出更高的愈合率,减少的炎症阶段和扩大的胶原蛋白安装。在其他研究中,将Au NP加载到N,N,N-三甲基壳聚糖/藻酸盐复合物中;这种纳米复合材料具有良好的生物相容性和高伤口敷料的前瞻性[186]。
虽然有许多方法可用于生产临床安全的NP,但它们的外在特性会对人体造成伤害。与各种药物治疗一样,过量暴露于NP可能导致不良副作用。例如,NPs分散和积聚到身体器官(例如脑,肺,肾和皮肤)中可以引发对宿主的毒性。为了避免这些风险,体内生物分布和安全降解特征研究对于非生物降解NP的临床应用至关重要[27]。此外,这些研究对于验证NP(保留在伤口敷料中)保留在皮肤层中,皮肤对NP的不良反应(例如炎症和异物反应)以及跟踪NP随后逃逸到血流中是至关重要的。
5.结论
由于感染引起的不愈合伤口仍然是一种难以治疗的,因此仍然可以设计可用作伤口敷料的先进材料。最近的发现允许生产伤口敷料,其有可能成为向伤口部位递送有益分子和/或药物的理想候选者。特别是,在本综述中,我们介绍了几种抗生素,精油,蜂蜜和无机纳米颗粒的掺入,最后一种允许产生复合材料用于多药理目标。
参考
1. van Koppen C.J., Hartmann R.W. Advances in the treatment of chronic wounds: A patent review. Expert Opin. Ther. Pat. 2015;25:931–937. doi: 10.1517/13543776.2015.1045879. [PubMed] [CrossRef]
2. Sorg H., Tilkorn D.J., Hager S., Hauser J., Mirastschijski U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017;58:81–94. doi: 10.1159/000454919. [PubMed] [CrossRef]
3. Bowler P.G., Duerden B.I., Armstrong D.G. Wound microbiology and associated approaches to wound management. Clin. Microbiol. Rev. 2001;14:244–269. doi: 10.1128/CMR.14.2.244-269.2001. [PMC free article] [PubMed] [CrossRef]
4. World Health Organization Who Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed. [(accessed on 26 May 2018)]; Available online: https://tinyurl.com/kmva5da.
5. Cardona A.F., Wilson S.E. Skin and soft-tissue infections: A critical review and the role of telavancin in their treatment. Clin. Infect. Dis. 2015;61:S69–S78. doi: 10.1093/cid/civ528. [PubMed] [CrossRef]
6. Vyas K., Vasconez H.C. Wound healing: Biologics, skin substitutes, biomembranes and scaffolds. Healthcare. 2014;2:356–400. doi: 10.3390/healthcare2030356. [PMC free article] [PubMed] [CrossRef]
7. Kopecki Z., Cowin A.J. Fighting chronic wound infection—One model at a time. Wound Pract. Res. J. Aust. Wound Manag. Assoc. 2017;25:6–13.
8. Jones V., Grey J.E., Harding K.G. Wound dressings. BMJ. 2006;332:777–780. doi: 10.1136/bmj.332.7544.777. [PMC free article] [PubMed] [CrossRef]
9. Mirza A. Nanomedicine and advanced technologies for burns: Preventing infection and facilitating wound healing. Adv. Drug Deliv. Rev. 2018;123:33–64. doi: 10.1016/j.addr.2017.08.001. [PMC free article] [PubMed] [CrossRef]
10. Ye S., Jiang L., Wu J., Su C., Huang C., Liu X., Shao W. Flexible amoxicillin-grafted bacterial cellulose sponges for wound dressing: In vitro and in vivo evaluation. ACS Appl. Mater. Interfaces. 2018;10:5862–5870. doi: 10.1021/acsami.7b16680. [PubMed] [CrossRef]
11. Anjum S., Arora A., Alam M.S., Gupta B. Development of antimicrobial and scar preventive chitosan hydrogel wound dressings. Int. J. Pharm. 2016;508:92–101. doi: 10.1016/j.ijpharm.2016.05.013. [PubMed] [CrossRef]
12. Anjum A., Sim C.H., Ng S.F. Hydrogels Containing Antibiofilm and Antimicrobial Agents Beneficial for Biofilm-Associated Wound Infection: Formulation Characterizations and In vitro Study. AAPS PharmSciTech. 2018;19:1219–1230. doi: 10.1208/s12249-017-0937-4. [PubMed] [CrossRef]
13. Rădulescu M., Holban A.M., Mogoantă L., Bălşeanu T.A., Mogoșanu G.D., Savu D., Popescu R.C., Fufă O., Grumezescu A.M., Bezirtzoglou E., et al. Fabrication, Characterization, and Evaluation of Bionanocomposites Based on Natural Polymers and Antibiotics for Wound Healing Applications. Molecules. 2016;21:761 doi: 10.3390/molecules21060761. [PubMed] [CrossRef]
14. Low W.L., Kenward K., Britland S.T., Amin M.C.I.M., Martin C. Essential oils and metal ions as alternative antimicrobial agents: A focus on tea tree oil and silver. Int. Wound J. 2017;14:369–384. doi: 10.1111/iwj.12611. [PubMed] [CrossRef]
15. Saporito F., Sandri G., Bonferoni M.C., Rossi S., Boselli C., Icaro Cornaglia A., Mannucci B., Grisoli P., Vigani B., Ferrari F. Essential oil-loaded lipid nanoparticles for wound healing. Int. J. Nanomed. 2017;2018:175–186. doi: 10.2147/IJN.S152529. [PMC free article] [PubMed] [CrossRef]
16. Ramasubbu D.A., Smith V., Hayden F., Cronin P. Systemic antibiotics for treating malignant wounds. Cochrane Database Syst. Rev. 2017;8:CD011609. doi: 10.1002/14651858.CD011609.pub2. [PubMed] [CrossRef]
17. Everts R. How to Treat Wound Infection. Prevention and Treatment. [(accessed on 26 May 2018)];2016 Available online: https://www.acc.co.nz/assets/pro ... und-infections.pdf.
18. Das P., Horton R. Antibiotics: Achieving the balance between access and excess. Lancet. 2016;387:102–104. doi: 10.1016/S0140-6736(15)00729-1. [PubMed] [CrossRef]
19. Aumeeruddy-Elalfi Z., Mahomoodally M. Chapter: Extraction techniques and pharmacological potential of essential oils from medicinal and aromatic plants of Mauritius. In: Peters M., editor. Essential Oils: Historical Significance, Chemical Composition and Medicinal Uses and Benefits. Nova Publisher; Hauppauge, NY, USA: 2016. pp. 51–80.
20. Aumeeruddy-Elalfi Z., Gurib-Fakim A., Mahomoodally M. Chemical composition, antimicrobial and antibiotic potentiating activity of essential oils from 10 tropical medicinal plants from Mauritius. J. Herb. Med. 2016;6:88–95. doi: 10.1016/j.hermed.2016.02.002. [CrossRef]
21. Scagnelli A.M. Therapeutic review: Manuka honey. J. Exot. Pet Med. 2016;25:168–171. doi: 10.1053/j.jepm.2016.03.007. [CrossRef]
22. Zarrintaj P., Moghaddam A.S., Manouchehri S., Atoufi Z., Amiri A., Amirkhani M.A., Nilforoushzadeh M.A., Saeb M.R., Hamblin M.R., Mozafari M. Can regenerative medicine and nanotechnology combine to heal wounds? The search for the ideal wound dressing. Nanomedicine. 2017;12:2403–2422. doi: 10.2217/nnm-2017-0173. [PubMed] [CrossRef]
23. McNamara K., Tofail S.A.M. Nanoparticles in biomedical applications. Adv. Phys. X. 2017;2:54–88. doi: 10.1080/23746149.2016.1254570. [CrossRef]
24. Zewde B., Ambaye A., Stubbs J., III, Raghavan D. A review of stabilized silver nanoparticles—Synthesis, biological properties, characterization, and potential areas of applications. JSM Nanotechnol. Nanomed. 2016;4:1043.
25. Cabuzu D., Cirja A., Puiu R., Grumezescu A.M. Biomedical applications of gold nanoparticles. Curr. Top. Med. Chem. 2015;15:1605–1613. doi: 10.2174/1568026615666150414144750. [PubMed] [CrossRef]
26. Pelgrift R.Y., Friedman A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013;65:1803–1815. doi: 10.1016/j.addr.2013.07.011. [PubMed] [CrossRef]
27. Yang Y., Qin Z., Zeng W., Yang T., Cao Y., Mei C., Kuang Y. Toxicity assessment of nanoparticles in various systems and organs. Nanotechnol. Rev. 2017;6:279–289. doi: 10.1515/ntrev-2016-0047. [CrossRef]
28. Chua A.W.C., Tan B.K., Foo C.L., Tan K.C., Chong S.J., Khoo Y.C. Skin tissue engineering advances in severe burns: Review and therapeutic applications. Burns Trauma. 2016;4:3–17. doi: 10.1186/s41038-016-0027-y. [PMC free article] [PubMed] [CrossRef]
29. Ho J., Walsh C., Yue D., Dardik A., Cheema U. Current Advancements and Strategies in Tissue Engineering for Wound Healing: A Comprehensive Review. Adv. Wound Care. 2017;6:191–209. doi: 10.1089/wound.2016.0723. [PMC free article] [PubMed] [CrossRef]
30. Olivier G., Wael N.H., Gamal B. Wound healing: Time to look for intelligent, ‘natural’ immunological approaches? BMC Immunol. 2017;18(Suppl. 1):23 doi: 10.1186/s12865-017-0207-y. [PMC free article] [PubMed] [CrossRef]
31. Enoch S., Leaper D.J. Basic science of wound healing. Surgery. 2008;26:31–37. doi: 10.1016/j.mpsur.2007.11.005. [CrossRef]
32. Xue M., Jackson C.J. Extracellular Matrix Reorganization during Wound Healing and Its Impact on Abnormal Scarring. Adv. Wound Care. 2015;4:119–136. doi: 10.1089/wound.2013.0485. [PMC free article] [PubMed] [CrossRef]
33. Rittié L. Cellular mechanisms of skin repair in humans and other mammals. J. Cell Commun. Signal. 2016;10:103–120. doi: 10.1007/s12079-016-0330-1. [PMC free article] [PubMed] [CrossRef]
34. Sanon S., Hart D.A., Tredget E.E. Molecular and cellular biology of wound healing and skin regeneration. In: Albanna M.Z., Holmes J.H., editors. Skin Tissue Engineering and Regenerative Medicine. Elsevier Inc.; New York, NY, USA: 2016. pp. 19–47.
35. Martin P., Nunan R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br. J. Dermatol. 2015;173:370–378. doi: 10.1111/bjd.13954. [PMC free article] [PubMed] [CrossRef]
36. Demidova-Rice T.N., Durham J.T., Herman I.M. Wound healing angio genesis: Innovations and challenges in acute and chronic wound healing. Adv. Wound Care. 2012;1:17–22. doi: 10.1089/wound.2011.0308. [PMC free article] [PubMed] [CrossRef]
37. Gould L.J., Fulton A.T. Wound Healing in Older Adults. R. I. Med. J. 2016;99:34–36. [PubMed]
38. Boateng S., Matthews K.H., Stevens H.N.E., Eccleston G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm. Sci. 2008;97:2892–2923. doi: 10.1002/jps.21210. [PubMed] [CrossRef]
39. van Rensburg J.J., Lin H., Gao X., Toh E., Fortney K.R., Ellinger S., Zwickl B., Janowicz D.M., Katz B.P., Nelson D.E., et al. The human skin microbiome associates with the outcome of and is influenced by bacterial infection. mBio. 2015;6:e01315-15. doi: 10.1128/mBio.01315-15. [PMC free article] [PubMed] [CrossRef]
40. Grice E.A. The skin microbiome: Potential for novel diagnostic and therapeutic approaches to cutaneous disease. Semin. Cutan. Med. Surg. 2014;33:98–103. doi: 10.12788/j.sder.0087. [PMC free article] [PubMed] [CrossRef]
41. Sarheed O., Ahmed A., Shouqair D., Boateng J. Antimicrobial dressings for improving wound healing. In: Alexandrescu V., editor. Wound Healing-New Insights into Ancient Challenges. InTech; London, UK: 2016. pp. 373–398.
42. Serra R., Grande R., Butrico L., Rossi A., Settimio U.F., Caroleo B., Amato B., Gallelli L., de Franciscis S. Chronic wound infections: The role of Pseudomonas aeruginosa and Staphylococcus aureus. Expert Rev. Anti-Infect. Ther. 2015;13:605–613. doi: 10.1586/14787210.2015.1023291. [PubMed] [CrossRef]
43. Guo S., Dipietro L.A. Factors affecting wound healing. J. Dent. Res. 2010;89:219–229. doi: 10.1177/0022034509359125. [PMC free article] [PubMed] [CrossRef]
44. Ortines R.V., Cheng L., Cohen T.S., Gami A., Dillen C.A., Ashbaugh A.G., Miller R.J., Wang Y., Tkaczyk C., Sellman B.R., et al. Anti-alpha-toxin immunoprohylaxis reduces disease severity against a Staphylococcus aureus full-thickness skin wound infection in immunocompetent and diabetic mice. [(accessed on 26 May 2018)];J. Immunol. 2017 198(Suppl. 1):77.20. Available online: http://www.jimmunol.org/content/198/1_Supplement/77.20.
45. Peerayeh S.N., Moghadas A.J., Behmanesh M. Prevalence of Virulence-Related Determinants in Clinical Isolates of Staphylococcus epidermidis. Jundishapur J. Microbiol. 2016;9:e30593. doi: 10.5812/jjm.30593. [PMC free article] [PubMed] [CrossRef]
46. Regev A., Weinberger M., Fishman M., Samra Z., Pitlik S.D. Necrotizing fasciitis caused by Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 1998;17:101–103. doi: 10.1007/s100960050025. [PubMed] [CrossRef]
47. Lu J., Yang M., Zhan M., Xu X., Yue J., Xu T. Antibiotics for treating infected burn wounds. Cochrane Database Syst. Rev. 2016;2:CD012084. doi: 10.1002/14651858.CD012084. [CrossRef]
48. Church D., Lloyd T., Peirano G., Pitout J. Antimicrobial susceptibility and combination testing of invasive Stenotrophomonas maltophilia isolates. Scand. J. Infect. Dis. 2013;45:265–270. doi: 10.3109/00365548.2012.732240. [PubMed] [CrossRef]
49. Brooke J.S. Stenotrophomonas maltophilia: An emerging global opportunistic pathogen. Clin. Microbiol. Rev. 2012;25:2–41. doi: 10.1128/CMR.00019-11. [PMC free article] [PubMed] [CrossRef]
50. Moet G.J., Jonesab R.N., Biedenbach D.J., Stilwell M.G., Fritsche T.R. Contemporary causes of skin and soft tissue infections in North America, Latin America, and Europe: Report from the SENTRY Antimicrobial Surveillance Program (1998–2004) Diagn. Microbiol. Infect. Dis. 2007;57:7–13. doi: 10.1016/j.diagmicrobio.2006.05.009. [PubMed] [CrossRef]
51. Kishore J. Isolation, identification & characterization of Proteus penneri—A missed rare pathogen. Indian J. Med. Res. 2012;135:341–345. [PMC free article] [PubMed]
52. Mihai M.M., Holban A.M., Giurcăneanu C., Popa L.G., Buzea M., Filipov M., Lazăr V., Chifiriuc M.C., Popa M.I. Identification and phenotypic characterization of the most frequent bacterial etiologies in chronic skin ulcers. Rom. J. Morphol. Embryol. 2014;55:1401–1408. [PubMed]
53. Lee M.J., Pottinger P.S., Butler-Wu S., Bumgarner R.E., Russ S.M., Matsen F.A. Propionibacterium persists in the skin despite standard surgical preparation. J. Bone Jt. Surg. Am. 2014;96:1447–1450. doi: 10.2106/JBJS.M.01474. [PubMed] [CrossRef]
54. Howard A., O’Donoghue M., Feeney A., Sleator R.D. Acinetobacter baumannii: An emerging opportunistic pathogen. Virulence. 2012;3:243–250. doi: 10.4161/viru.19700. [PMC free article] [PubMed] [CrossRef]
55. Rotstein O.D., Vittorini T., Kao J., McBurney M.I., Nasmith P.E., Grinstein S. A soluble Bacteroides by-product impairs phagocytic killing of Escherichia coli by neutrophils. Infect. Immun. 1989;57:745–753. [PMC free article] [PubMed]
56. Cutting K.F., White R.J. Criteria for identifying wound infection revisited. Ostomy Wound Manag. 2005;51:28–34. [PubMed]
57. Felk A., Kretschmar M., Albrecht A., Schaller M., Beinhauer S., Nichterlein T., Sanglard D., Korting H.C., Schäfer W., Hube B. Candida albicans hyphal formation and the expression of the Efg1-regulated proteinases Sap4 to Sap6 are required for the invasion of parenchymal organs. Infect. Immun. 2002;70:3689–3700. doi: 10.1128/IAI.70.7.3689-3700.2002. [PMC free article] [PubMed] [CrossRef]
58. Katakura T., Yoshida T., Kobayashi M., Herndon D.N., Suzuki F. Immunological control of methicillin-resistant Staphylococcus aureus (MRSA) infection in an immunodeficient murine model of thermal injuries. Clin. Exp. Immunol. 2005;142:419–425. doi: 10.1111/j.1365-2249.2005.02944.x. [PMC free article] [PubMed] [CrossRef]
59. Shirtliff M.E., Peters B.M., Jabra-Rizk M.A. Cross-kingdom interactions: Candida albicans and bacteria. FEMS Microbiol. Lett. 2009;299:1–8. doi: 10.1111/j.1574-6968.2009.01668.x. [PMC free article] [PubMed] [CrossRef]
60. Beele H., Meuleneire F., Nahuys M., Percival S.L. A prospective randomised open label study to evaluate the potential of a new silver alginate/carboxymethylcellulose antimicrobial wound dressing to promote wound healing. Int. Wound J. 2010;7:262–270. doi: 10.1111/j.1742-481X.2010.00669.x. [PubMed] [CrossRef]
61. Percival S.L., Bowler MPhil P., Woods E.J. Assessing the effect of an antimicrobial wound dressing on biofilms. Wound Repair Regen. 2008;16:52–57. doi: 10.1111/j.1524-475X.2007.00350.x. [PubMed] [CrossRef]
62. Koehler J., Brand F.P., Goepferich A.M. Hydrogel wound dressings for bioactive treatment of acute and chronic wounds. Eur. Polym. J. 2018;100:1–11. doi: 10.1016/j.eurpolymj.2017.12.046. [CrossRef]
63. Dutra J.A.P., Carvalho S.G., Zampirolli A.C.D., Daltoé R.D., Teixeira R.M., Careta F.P., Cotrim M.A.P., Oréfice R.L., Villanova J.C.O. Papain wound dressings obtained from poly (vinyl alcohol)/calcium alginate blends as new pharmaceutical dosage form: Preparation and preliminary evaluation. Eur. J. Pharm. Biopharm. 2017;113:11–23. doi: 10.1016/j.ejpb.2016.12.001. [PubMed] [CrossRef]
64. Sasikala L., Dhurai B. Preparation and Analysis of Chitosan-Honey Films for Wound Dressing Application. [(accessed on 26 May 2018)];World Acad. Sci. Eng. Technol. Int. J. Mater. Text. Eng. 2018 12:54. Available online: urn:dai:10.1999/1307-6892/75464.
65. Ahmed A., Boateng J. Calcium alginate-based antimicrobial film dressings for potential healing of infected foot ulcers. Ther. Deliv. 2018;9:185–204. doi: 10.4155/tde-2017-0104. [PubMed] [CrossRef]
66. Capanema N.S.V., Mansur A.A.P., Carvalho S.M., Mansur L.L., Ramos C.P., Lage A.P., Mansur H.S. Physicochemical properties and antimicrobial activity of biocompatible carboxymethylcellulose-silver nanoparticle hybrids for wound dressing and epidermal repair. J. Appl. Polym. Sci. 2018;135:45812. doi: 10.1002/app.45812. [CrossRef]
67. Yao C.H., Lee C.Y., Huang C.H., Chen Y.S., Chen K.Y. Novel bilayer wound dressing based on electrospun gelatin/keratin nanofibrous mats for skin wound repair. Mater. Sci. Eng. C-Mater. Biol. Appl. 2017;79:533–540. doi: 10.1016/j.msec.2017.05.076. [PubMed] [CrossRef]
68. Cerchiara T., Abruzzo A., Ñahui Palomino R.A., De Rose B.V.R., Chidichimo G., Ceseracciu L., Athanassiou A., Saladini B., Dalena F., Bigucci F., et al. Spanish Broom (Spartium junceum L.) fibers impregnated with vancomycin-loaded chitosan nanoparticles as new antibacterial wound dressing: Preparation, characterization and antibacterial activity. Eur. J. Pharm. Sci. 2017;99:105–112. doi: 10.1016/j.ejps.2016.11.028. [PubMed] [CrossRef]
69. Swenty C.F. Principles to Guide Your Dressing Choice. J. Nurse Pract. 2016;12:e125–e127. doi: 10.1016/j.nurpra.2015.12.006. [CrossRef]
70. Asfaw T., Jackson J.C., Lu Z., Zhai X., Shums S., Hirt T., Hu X., René C.R. In-Situ Forming Hydrogel Wound Dressings Containing Antimicrobial Agents. US923280B2. U.S. Patent. 2016 Jan 12;
71. Ousey K., Cutting K., Rogers A.A., Rippon M. The importance of hydration in wound healing: Reinvigorating the clinical perspective. J. Wound Care. 2016;25:122–130. doi: 10.12968/jowc.2016.25.3.122. [PubMed] [CrossRef]
72. Dhivya S., Padma V.V., Santhini E. Wound dressings—A review. Biomedicine. 2015;5:24–28. doi: 10.7603/s40681-015-0022-9. [PMC free article] [PubMed] [CrossRef]
73. Qiu Y., Qiu L., Cui J., Wei Q. Bacterial cellulose and bacterial cellulose-vaccarin membranes for wound healing. Mater. Sci. Eng. C. 2016;59:303–309. doi: 10.1016/j.msec.2015.10.016. [PubMed] [CrossRef]
74. Fan L., Yang H., Yang J., Peng M., Hu J. Preparation and characterization of chitosan/gelatin/PVA hydrogel for wound dressings. Carbohydr. Polym. 2016;146:427–434. doi: 10.1016/j.carbpol.2016.03.002. [PubMed] [CrossRef]
75. Dabiri G., Damstetter E., Phillips T. Choosing a wound dressing based on common wound characteristics. Adv. Wound Care. 2016;5:32–41. doi: 10.1089/wound.2014.0586. [PMC free article] [PubMed] [CrossRef]
76. Ramos-e-Silva M., de Castro M.C.R. New dressings, including tissue-engineered living skin. Clin. Dermatol. 2002;20:715–723. doi: 10.1016/S0738-081X(02)00298-5. [PubMed] [CrossRef]
77. Flores C., Lopez M., Tabary N., Neut C., Chai F., Betbeder D., Herkt C., Cazaux F., Gaucher V., Martel B. Preparation and characterization of novel chitosan and β-cyclodextrin polymer sponges for wound dressing applications. Carbohydr. Polym. 2017:535–546. doi: 10.1016/j.carbpol.2017.06.026. [PubMed] [CrossRef]
78. Pott F.S., Meier M.J., Stocco J.G.D., Crozeta K., Ribas J.D. The effectiveness of hydrocolloid dressings versus other dressings in the healing of pressure ulcers in adults and older adults: A systematic review and meta-analysis. Rev. Lat.-Am. Enferm. 2014;22:511–529. doi: 10.1590/0104-1169.3480.2445. [PMC free article] [PubMed] [CrossRef]
79. Das S., Baker A. Biomaterials and Nanotherapeutics for Enhancing Skin Wound Healing. Front. Bioeng. Biotechnol. 2016;4:82. doi: 10.3389/fbioe.2016.00082. [PMC free article] [PubMed] [CrossRef]
80. Woundcarehandbook. [(accessed on 30 June 2018)]; Available online: http://www.woundcarehandbook.com.
81. Sweeney I.R., Miraftab M., Collyer G. A critical review of modern and emerging absorbent dressings used to treat exuding wounds. Int. Wound J. 2012;9:601–612. doi: 10.1111/j.1742-481X.2011.00923.x. [PubMed] [CrossRef]
82. Chattopadhyay S., Raines R.T., Glick G.D. Review collagen-based biomaterials for wound healing. Biopolymers. 2014;101:821–833. doi: 10.1002/bip.22486. [PMC free article] [PubMed] [CrossRef]
83. Calo E., Ballamy L., Khutoryanskiy V.V. Hydrogels in Wound Management. In: Singh T.R.R., Leverty G., Donelly R., editors. Hydrogels: Design, Synthesis and Application in Drug Delivery and Regenerative Medicine. CRC Press; Boca Raton, FL, USA: 2018.
84. Vermeulen H., Ubbink D.T., Goossens A., de Vos R., Legemate D.A. Systematic review of dressings and topical agents for surgical wounds healing by secondary intention. Br. J. Surg. 2005;92:665–672. doi: 10.1002/bjs.5055. [PubMed] [CrossRef]
85. International Wound Infection Institute (IWII) In: Wound Infection in Clinical Practice. Terry Swanson N.P.W.M., editor. Wounds International; London, UK: 2016. [(accessed on 25 July 2018)]. Available online: https://tinyurl.com/y8skcrnd.
86. Liu X., Nielsen L.H., Kłodzińska S.N., Nielsen H.M., Quc H., Christensen L.P., Rantanen J., Yangad M. Ciprofloxacin-loaded sodium alginate/poly(lactic-co-glycolic acid) electrospun fibrous mats for wound healing. Eur. J. Pharm. Biopharm. 2018;123:42–49. doi: 10.1016/j.ejpb.2017.11.004. [PubMed] [CrossRef]
87. Contardi M., Heredia-Guerrero J.A., Perotto G., Valentini P., Pompa P.P., Spanò R., Goldonic L., Bertorelli R., Athanassiou A., Bayera I.S. Transparent ciprofloxacin-povidone antibiotic films and nanofiber mats as potential skin and wound care dressings. Eur. J. Pharm. Sci. 2017;104:133–144. doi: 10.1016/j.ejps.2017.03.044. [PubMed] [CrossRef]
88. Li H., Williams G.R., Wang J.W.H., Sun X., Zhu L.M. Poly(N-isopropylacrylamide)/poly(l-lactic acid-co-ɛ-caprolactone) fibers loaded with ciprofloxacin as wound dressing materials. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;79:245–254. doi: 10.1016/j.msec.2017.04.058. [PubMed] [CrossRef]
89. Pamfil D., Vasile C., Tarţău L., Vereştiuc L., Poiată A. pH-Responsive 2-hydroxyethyl methacrylate/citraconic anhydride–modified collagen hydrogels as ciprofloxacin carriers for wound dressings. J. Bioact. Compat. Polym. 2017;32:355–381. doi: 10.1177/0883911516684653. [CrossRef]
90. Khampieng T., Wnek G.E., Supaphol P. Electrospun DOXY-h loaded-poly(acrylic acid) nanofiber mats: In vitro drug release and antibacterial properties investigation. J. Biomater. Sci.-Polym. Ed. 2014;25:1292–1305. doi: 10.1080/09205063.2014.929431. [PubMed] [CrossRef]
91. Michalska-Sionkowska M., Kaczmarek B., Walczak M., Sionkowska A. Antimicrobial activity of new materials based on the blends of collagen/chitosan/hyaluronic acid with gentamicin sulfate addition. Mater. Sci. Eng. C Mater. Biol. Appl. 2018;86:103–108. doi: 10.1016/j.msec.2018.01.005. [PubMed] [CrossRef]
92. Ahire J.J., Robertson D.D., van Reenen A.J., Dicks L.M.T. Polyethylene oxide (PEO)-hyaluronic acid (HA) nanofibers with kanamycin inhibits the growth of Listeria monocytogenes. Biomed. Pharmacother. 2017;86:143–148. doi: 10.1016/j.biopha.2016.12.006. [PubMed] [CrossRef]
93. Etebu E., Arikekpar I. Antibiotics: Classification and mechanisms of action with emphasis on molecular perspectives. Int. J. Appl. Microbiol. Biotechnol. Res. 2016;4:90–101.
94. Kohanski M.A., Dwyer D.J., Collins J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010;8:423–435. doi: 10.1038/nrmicro2333. [PMC free article] [PubMed] [CrossRef]
95. Rai M., Kon K., Gade A., Ingle A., Nagaonkar D., Paralikar P., da Silva S.S. Antibiotic Resistance. Mechanisms and New Antimicrobial Approaches. Elsevier Science; New York, NY, USA: 2016. Chapter 6—Antibiotic Resistance: Can Nanoparticles Tackle the Problem? pp. 121–143. [CrossRef]
96. Friedman N.D., Temkin E., Carmeli Y. The negative impact of antibiotic resistance. Clin. Microbiol. Infect. 2016;22:416–422. doi: 10.1016/j.cmi.2015.12.002. [PubMed] [CrossRef]
97. Pîrvănescu H., Bălăşoiu M., Ciurea M.E., Bălăşoiu A.T., Mănescu R. Wound infections with multi-drug resistant bacteria. Chirurgia. 2014;109:73–79. [PubMed]
98. Chávez-González M.L., Rodríguez-Herrera R., Aguilar C.N. Antibiotic Resistance. Mechanisms and New Antimicrobial Approaches. Elsevier Science; New York, NY, USA: 2016. Chapter 11—Essential Oils: A Natural Alternative to Combat Antibiotics Resistance; pp. 227–237. [CrossRef]
99. Shrestha G., Raphael J., Leavitt S.D., St Clair L.L. In vitro evaluation of the antibacterial activity of extracts from 34 species of North American lichens. Pharm. Biol. 2014;52:1262–1266. doi: 10.3109/13880209.2014.889175. [PubMed] [CrossRef]
100. Segev-Zarko L., Saar-Dover R., Brumfeld V., Mangoni M.L., Shai Y. Mechanisms of biofilm inhibition and degradation by antimicrobial peptides. Biochem. J. 2015;468:259–270. doi: 10.1042/BJ20141251. [PubMed] [CrossRef]
101. Seow Y.X., Yeo C.R., Chung H.L., Yuk H.-G. Plant essential oils as active antimicrobialagents. Crit. Rev. Food Sci. Nutr. 2014;54:625–644. doi: 10.1080/10408398.2011.599504. [PubMed] [CrossRef]
102. Agyare C., Duah Y., Oppong E., Hensel A., Oteng S., Appiah T. Review: African medicinal plants with wound healing properties. J. Ethnopharmacol. 2016;177:85–100. doi: 10.1016/j.jep.2015.11.008. [PubMed] [CrossRef]
103. Semeniuc C.A., Popa C.R., Rotar A.M. Antibacterial activity and interactions of plant essential oil combinations againts Gram-positive and Gram-negative bacteria. J. Food Drug Anal. 2017;25:403–408. doi: 10.1016/j.jfda.2016.06.002. [PubMed] [CrossRef]
104. Kavoosi G., Dadfar S.M.M., Purfard A.M., Mehrabi R. Antioxidant and Antibacterial Properties of Gelatin Films Incorporated with Carvacrol. J. Food Saf. 2013;33:423–432. doi: 10.1111/jfs.12071. [CrossRef]
105. Altiok D., Altiok E., Tihminlioglu F. Physical, antibacterial and antioxidant properties of chitosan films incorporated with thyme oil for potential wound healing applications. J. Mater. Sci. Mater. Med. 2010;21:2227–2236. doi: 10.1007/s10856-010-4065-x. [PubMed] [CrossRef]
106. Walsh S.E., Maillard J.-Y., Russell A.D., Catrenich C.E., Charbonneau D.L., Bartolo R.G. Development of bacterial resistance to several biocides and effects on antibiotic susceptibility. J. Hosp. Infect. 2003;55:98–107. doi: 10.1016/S0195-6701(03)00240-8. [PubMed] [CrossRef]
107. Sienkiewicz M., Głowacka A., Kowalczyk E., Wiktorowska-Owczarek A., Jóźwiak-Bębenista M., Łysakowska M. The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils. Molecules. 2014;19:20929–20940. doi: 10.3390/molecules191220929. [PMC free article] [PubMed] [CrossRef]
108. Zenati F., Benbelaid F., Khadir A., Bellahsene C., Bendahou M. Antimicrobial effects of three essential oils on multidrug resistant bacteria responsible for urinary infections. J. Appl. Pharm. Sci. 2014;4:15–18. doi: 10.7324/JAPS.2014.4113. [CrossRef]
109. Liakos I., Rizzello L., Scurr D.J., Pompa P.P., Bayer I.S., Athanassiou A. All-natural composite wound dressing films of essential oils encapsulated in sodium alginate with antimicrobial properties. Int. J. Pharm. 2014;463:137–145. doi: 10.1016/j.ijpharm.2013.10.046. [PubMed] [CrossRef]
110. Liakos I., Rizzello L., Hajiali H., Brunetti V., Carzino R., Pompa P., Athanassiou A., Mele E. Fibrous wound dressings encapsulating essential oils as natural antimicrobial agents. J. Mater. Chem. B. 2015;3:1583–1589. doi: 10.1039/C4TB01974A. [CrossRef]
111. Rosa J.M., Bicudo Bonato L., Bragine Mancuso C., Martinelli L., Okura M.H., Malpass G.R.P., Granato A.C. Antimicrobial wound dressing films containing essential oils and oleoresins of pepper encapsulated in sodium alginate films. Cienc. Rural. 2018;48:e20170740. doi: 10.1590/0103-8478cr20170740. [CrossRef]
112. Nogueira M.N.M., Aquino S.G., Rossa Junior C., Spolidorio D.M.P. Terpinen-4-ol and alpha-terpineol (tea tree oil components) inhibit the production of IL-1b, IL-6 and IL-10 on human macrophages. Inflamm. Res. 2014;63:769–778. doi: 10.1007/s00011-014-0749-x. [PubMed] [CrossRef]
113. Bai M.-Y., Chou T.-C., Tsai J.-C., Yu W.-C. The effect of active ingredient-containing chitosan/polycaprolactone nonwoven mat on wound healing: In vitro and in vivo studies. J. Biomed. Mater. Res. Part A. 2014;102:2324–2333. doi: 10.1002/jbm.a.34912. [PubMed] [CrossRef]
114. Ge Y., Ge M. Sustained broad-spectrum antimicrobial and haemostatic chitosan-based film with immerged tea tree oil droplets. Fibers Polym. 2015;16:308–318. doi: 10.1007/s12221-015-0308-2. [CrossRef]
115. Edmondson M., Newall N., Carville K., Smith J., Riley T.V., Carson C.F. Uncontrolled, open-label, pilot study of tea tree (Melaleuca alternifolia) oil solution in the decolonisation of methicillin-resistant Staphylococcus aureus positive wounds and its influence on wound healing. Int. Wound J. 2011;8:375–384. doi: 10.1111/j.1742-481X.2011.00801.x. [PubMed] [CrossRef]
116. Lee R., Leung P., Wong T. A randomized controlled trial of topical tea tree preparation for MRSA colonized wounds. Int. J. Nurs. Sci. 2014;1:7–14. doi: 10.1016/j.ijnss.2014.01.001. [CrossRef]
117. Cuttle L., Kempf M., Kravchuk O., George N., Liu P.Y., Chang H.E., Mill J., Wang X.Q., Kimble R.M. The efficacy of Aloe vera, tea tree oil and saliva as first aid treatment for partial thickness burn injuries. Burns. 2008;34:1176–1182. doi: 10.1016/j.burns.2008.03.012. [PubMed] [CrossRef]
118. Saddiqe Z., Naeem I., Maimoona A. A review of the antibacterial activity of Hypericum perforatum L. J. Ethnopharmacol. 2010;131:511–521. doi: 10.1016/j.jep.2010.07.034. [PubMed] [CrossRef]
119. Güneş S., Tıhmınlıoğlu F. Hypericum perforatum incorporated chitosan films as potential bioactive wound dressing material. Int. J. Biol. Macromol. 2017;102:933–943. doi: 10.1016/j.ijbiomac.2017.04.080. [PubMed] [CrossRef]
120. Evandri M.G., Battinelli L., Daniele C., Mastrangelo S., Bolle P., Mazzanti G. The antimutagenic activity of Lavandula angustifolia (lavender) essential oil in the bacterial reverse mutation assay. Food Chem. Toxicol. 2005;43:1381–1387. doi: 10.1016/j.fct.2005.03.013. [PubMed] [CrossRef]
121. Cavanagh H.M.A., Wilkinson J.M. Biological activities of lavender essentials oil. Phytother. Res. 2002;16:301–308. doi: 10.1002/ptr.1103. [PubMed] [CrossRef]
122. Imane M.M., Houda F., Amal A.H.S., Kaotar N., Mohammed T., Imane R., Farid H. Phytochemical Composition and Antibacterial Activity of Moroccan Lavandula angustifolia Mill. J. Essent. Oil Bear. Plants. 2017;20:1074–1082. doi: 10.1080/0972060X.2017.1363000. [CrossRef]
123. Mori H., Kawanami H., Kawahata H., Aoki M. Wound healing potential of lavender oil by acceleration of granulation and wound contraction through induction of TGF-β in a rat model. BMC Complement. Altern. Med. 2016;16:144 doi: 10.1186/s12906-016-1128-7. [PMC free article] [PubMed] [CrossRef]
124. Sarikurkcu C., Zengin G., Oskay M., Uysal S., Ceylan R., Aktumsek A. Composition, antioxidant, antimicrobial and enzyme inhibition activities of two Origanum vulgare subspecies (subsp. vulgare and subsp. hirtum) essential oils. Ind. Crops Prod. 2015;70:178–184. doi: 10.1016/j.indcrop.2015.03.030. [CrossRef]
125. Nostro A., Blanco A.R., Cannatelli M.A., Enea V., Flamini G., Morelli I., Sudano Roccaro A., Alonzo V. Susceptibility of methicillin-resistant staphylococci to oregano essential oil, carvacrol and thymol. FEMS Microbiol. Lett. 2004;230:191–195. doi: 10.1016/S0378-1097(03)00890-5. [PubMed] [CrossRef]
126. Liakos I.L., Holban A.M., Carzino R., Lauciello S., Grumezescu A.M. Electrospun Fiber Pads of Cellulose Acetate and Essential Oils with Antimicrobial Activity. Nanomaterials. 2017;7:84 doi: 10.3390/nano7040084. [PMC free article] [PubMed] [CrossRef]
127. Bogdanov S. The Bee Products: The Wonders of the Bee Hexagon, Bee Product Science. [(accessed on 1 August 2018)]; Available online: www.bee-hexagon.net.
128. Kwakman P.H., te Velde A.A., de Boer L., Speijer D., Vandenbroucke-Grauls C.M., Zaat S.A. How honey kills bacteria. FASEB J. 2010;24:2576–2582. doi: 10.1096/fj.09-150789. [PubMed] [CrossRef]
129. Molan P.C. The evidence supporting the use of honey as a wound dressing. Int. J. Lower Extrem. Wounds. 2006;5:40–54. doi: 10.1177/1534734605286014. [PubMed] [CrossRef]
130. Israili Z.H. Antimicrobial properties of honey. Am. J. Ther. 2014;21:304–423. doi: 10.1097/MJT.0b013e318293b09b. [PubMed] [CrossRef]
131. Boateng J., Diunase K.N. Comparing the antibacterial and functional properties of cameroonian and manuka honeys for potential wound healing—Have we come full cycle in dealing with antibiotic resistance? Molecules. 2015;20:16068–16084. doi: 10.3390/molecules200916068. [PMC free article] [PubMed] [CrossRef]
132. Simon A., Traynor K., Santos K., Blaser G., Bode U., Molan P. Medical honey for wound care—Still the ‘latest resort’? Evid.-Based Complement. Altern. Med. 2009;6:165–173. doi: 10.1093/ecam/nem175. [PMC free article] [PubMed] [CrossRef]
133. Kuś P.M., Szweda P., Jerković I., Tuberoso C.I. Activity of Polish unifloral honeys against pathogenic bacteria and its correlation with colour, phenolic content, antioxidant capacity and other parameters. Lett. Appl. Microbiol. 2016;62:269–276. doi: 10.1111/lam.12541. [PubMed] [CrossRef]
134. Sherlock O., Dolan A., Athman R., Power A., Gethin G., Cowman S., Humphreys H. Comparison of the antimicrobial activity of Ulmo honey from Chile and Manuka honey against methicillin-resistant Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. BMC Complement. Altern. Med. 2010;10:47 doi: 10.1186/1472-6882-10-47. [PMC free article] [PubMed] [CrossRef]
135. Ng W.J., Lim M.S. Anti-staphylococcal activity of melaleuca honey. Southeast Asian J. Trop. Med. Public Health. 2015;46:472–479. [PubMed]
136. Jantakee K., Tragoolpua Y. Activities of different types of Thai honey on pathogenic bacteria causing skin diseases, tyrosinase enzyme and generating free radicals. Biol. Res. 2015;48:4. doi: 10.1186/0717-6287-48-4. [PMC free article] [PubMed] [CrossRef]
137. Packer J.M., Irish J., Herbert B.R., Hill C., Padula M., Blair S.E., Carter D.A., Harry E.J. Specific non-peroxide antibacterial effect of manuka honey on the Staphylococcus aureus proteome. Int. J. Antimicrob. Agents. 2012;40:43–50. doi: 10.1016/j.ijantimicag.2012.03.012. [PubMed] [CrossRef]
138. Cooper R. Honey as an effective antimicrobial treatment for chronic wounds: Is there a place for it in modern medicine? Chronic Wound Care Manag. Res. 2014;1:15–22. doi: 10.2147/CWCMR.S46520. [CrossRef]
139. Bulman S.E., Tronci G., Goswami P., Carr C., Russell S.J. Antibacterial properties of nonwoven wound dressings coated with Manuka honey or methylglyoxal. Materials. 2017;10:954 doi: 10.3390/ma10080954. [PMC free article] [PubMed] [CrossRef]
140. Lu J., Turnbull L., Burke C.M., Liu M., Carter D.A., Schlothauer R.C., Whitchurch C.B., Harry E.J. Manuka-type honeys can eradicate biofilms produced by Staphylococcus aureus strains with different biofilm-forming abilities. PeerJ. 2014;2:e326. doi: 10.7717/peerj.326. [PMC free article] [PubMed] [CrossRef]
141. Minden-Birkenmaier B.A., Neuhalfen R.M., Janowiak B.E., Sell S.A. Preliminary Investigation and Characterization of Electrospun Polycaprolactone and Manuka Honey Scaffolds for Dermal Repair. J. Eng. Fiber Fabr. 2015;10:126–138.
142. Yang X., Fan L., Ma L., Wang Y., Lin S., Yu F., Pan X., Luo G., Zhang D., Wang H. Green electrospun Manuka honey/silk fibroin fibrous matrices as potential wound dressing. Mater. Des. 2017;119:76–84. doi: 10.1016/j.matdes.2017.01.023. [CrossRef]
143. Tavakoli J., Tang Y. Honey/PVA hybrid wound dressings with controlled release of antibiotics: Structural, physico-mechanical and in-vitro biomedical studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;77:318–325. doi: 10.1016/j.msec.2017.03.272. [PubMed] [CrossRef]
144. Saikaly S.K., Khachemoune A. Honey and Wound Healing: An Update. Am. J. Clin. Dermatol. 2017;18:237–251. doi: 10.1007/s40257-016-0247-8. [PubMed] [CrossRef]
145. Rai M., Yadav A., Gade A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009;27:76–83. doi: 10.1016/j.biotechadv.2008.09.002. [PubMed] [CrossRef]
146. Kumar M., Curtis A., Hoskins C. Application of Nanoparticle Technologies in the Combat against Anti-Microbial Resistance. Pharmaceutics. 2018;10:11 doi: 10.3390/pharmaceutics10010011. [PMC free article] [PubMed] [CrossRef]
147. Kandi V., Kandi S. Antimicrobial properties of nanomolecules: Potential candidates as antibiotics in the era of multi-drug resistance. Epidemiol. Health. 2015;37:e2015020. doi: 10.4178/epih/e2015020. [PMC free article] [PubMed] [CrossRef]
148. Simões D., Miguel S.P., Ribeiro M.P., Coutinho P., Mendonça A.G., Correia I.J. Recent advances on antimicrobial wound dressing: A review. Eur. J. Pharm. Biopharm. 2018;127:130–141. doi: 10.1016/j.ejpb.2018.02.022. [PubMed] [CrossRef]
149. Wang L., Hu C., Shao L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017;12:1227–1249. doi: 10.2147/IJN.S121956. [PMC free article] [PubMed] [CrossRef]
150. Baek Y.W., An Y.J. Microbial toxicity of metal oxide nanoparticles (CuO, NiO, ZnO, and Sb2O3) to Escherichia coli, Bacillus subtilis, and Streptococcus aureus. Sci. Total Environ. 2011;409:1603–1608. doi: 10.1016/j.scitotenv.2011.01.014. [PubMed] [CrossRef]
151. Ashkarran A.A., Ghavami M., Aghaverdi H., Stroeve P., Mahmoudi M. Bacterial effects and protein corona evaluations: Crucial ignored factors for prediction of bio-efficacy of various forms of silver nanoparticles. Chem. Res. Toxicol. 2012;25:1231–1242. doi: 10.1021/tx300083s. [PubMed] [CrossRef]
152. Lu C., Brauer M.J., Botstein D. Slow growth induces heat-shock resistance in normal and respiratory-deficient yeast. Mol. Biol. Cell. 2009;20:891–903. doi: 10.1091/mbc.e08-08-0852. [PMC free article] [PubMed] [CrossRef]
153. Pramanik A., Laha D., Bhattacharya D., Pramanik P., Karmakar P. A novel study of antibacterial activity of copper iodide nanoparticle mediated by DNA and membrane damage. Colloids Surf. B. 2012;96:50–55. doi: 10.1016/j.colsurfb.2012.03.021. [PubMed] [CrossRef]
154. Rai M.K., Deshmukh S.D., Ingle A.P., Gade A.K. Silver nanoparticles, the powerful nanoweapon against multidrug-resistant bacteria. J. Appl. Microbiol. 2012;112:841–852. doi: 10.1111/j.1365-2672.2012.05253.x. [PubMed] [CrossRef]
155. Anisha B.S., Biswas R., Chennazhi K.P., Jayakumar R. Chitosan-hyaluronic acid/nano silver composite sponges for drug resistant bacteria infected diabetic wounds. Int. J. Biol. Macromol. 2013;62:310–320. doi: 10.1016/j.ijbiomac.2013.09.011. [PubMed] [CrossRef]
156. Nam G., Rangasamy S., Purushothaman B., Song J.M. The application of bactericidal silver nanoparticles in wound treatment. Nanomater. Nanotechnol. 2015;5:23–37. doi: 10.5772/60918. [CrossRef]
157. Verma J., Kanoujia J., Parashar P., Tripathi C.B., Saraf S.A. Wound healing applications of sericin/chitosan-capped silver nanoparticles incorporated hydrogel. Drug Deliv. Transl. Res. 2017;7:77–88. doi: 10.1007/s13346-016-0322-y. [PubMed] [CrossRef]
158. Kaba S.I., Egorova E.M. In vitro studies of the toxic effects of silver nanoparticles on HeLa and U937 cells. Nanotechnol. Sci. Appl. 2015;8:19–29. doi: 10.2147/NSA.S78134. [PMC free article] [PubMed] [CrossRef]
159. Zhou Y., Chen R., He T., Xu K., Du D., Zhao N., Cheng X., Yang J., Shi H., Lin Y. Biomedical potential of ultrafine Ag/AgCl nanoparticles coated on graphene with special reference to antimicrobial performances and burn wound healing. ACS Appl. Mater. Interfaces. 2016;8:15067–15075. doi: 10.1021/acsami.6b03021. [PubMed] [CrossRef]
160. Frankova J., Pivodova V., Vagnerova H., Juranova J., Ulrichova J. Effects of silver nanoparticles on primary cell cultures of fibroblasts and keratinocytes in a wound-healing model. J. Appl. Biomater. Funct. Mater. 2016;14:137–142. doi: 10.5301/jabfm.5000268. [PubMed] [CrossRef]
161. El-Naggar M.Y., Gohar Y.M., Sorour M.A., Waheeb M.G. Hydrogel dressing with a nano-formula against methicillin-resistant Staphylococcus aureus and Pseudomonas aeruginosa diabetic foot bacteria. J. Microbiol. Biotechnol. 2016;26:408–420. doi: 10.4014/jmb.1506.06048. [PubMed] [CrossRef]
162. Jaiswal M., Koul V., Dinda A.K. In vitro and in vivo investigational studies of a nanocomposite-hydrogel-based dressing with a silver-coated chitosan wafer for full-thickness skin wounds. J. Appl. Polym. Sci. 2016;133:43472. doi: 10.1002/app.43472. [CrossRef]
163. Nešović K., Kojić V., Rhee K.Y., Mišković-Stanković V. Electrochemical synthesis and characterization of silver doped poly(vinyl alcohol)/chitosan hydrogels. Corrosion. 2017;73:1437–1447. doi: 10.5006/2507. [CrossRef]
164. Hanif M., Juluri R.R., Fojan P., Popok V.N. Polymer films with size-selected silver nanoparticles as plasmon resonance-based transducers for protein sensing. Biointerface Res. Appl. Chem. 2016;6:1564–1568.
165. Higa A.M., Mambrini G.P., Hausen M., Strixino F.T., Leite F.L. Ag-nanoparticle-based nano-immunosensor for anti-glutathione s-transferase detection. Biointerface Res. Appl. Chem. 2016;6:1053–1058.
166. Wu J., Zheng Y., Song W., Luan J., Wen X., Wu Z., Chen X., Wang Q., Guo S. In situ synthesis of silver-nanoparticles/bacterial cellulose composites for slow-released antimicrobial wound dressing. Carbohydr. Polym. 2014;102:762–771. doi: 10.1016/j.carbpol.2013.10.093. [PubMed] [CrossRef]
167. Radulescu M., Andronescu E., Dolete G., Popescu R.C., Fufă O., Chifiriuc M.C., Mogoantă L., Bălşeanu T.A., Mogoşanu G.D., Grumezescu A.M., et al. Silver Nanocoatings for Reducing the Exogenous Microbial Colonization of Wound Dressings. Materials. 2016;9:345 doi: 10.3390/ma9050345. [PMC free article] [PubMed] [CrossRef]
168. Hajipour M.J., Fromm K.M., Ashkarran A.A., de Aberasturi D.J., de Larramendi I.R., Rojo T., Serpooshan V., Parak W.J., Mahmoudi M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012;30:499–511. doi: 10.1016/j.tibtech.2012.06.004. [PubMed] [CrossRef]
169. Shafiee M.R.M., Kargar M. Synthesis of 3,4,5-substituted furan-2(5h)-ones using zno nanostructure as an efficient catalyst. Biointerface Res. Appl. Chem. 2017;7:2170–2173.
170. Applerot G., Lellouche J., Perkas N., Nitzan Y., Gedanken A., Banin E. ZnO nanoparticle-coated surfaces inhibit bacterial biofilm formation and increase antibiotic susceptibility. RSC Adv. 2012;2:2314–2321. doi: 10.1039/c2ra00602b. [CrossRef]
171. Mirzaei H., Darroudi M. Zinc oxide nanoparticles: Biological synthesis and biomedical applications. Ceram. Int. 2017;43:907–914. doi: 10.1016/j.ceramint.2016.10.051. [CrossRef]
172. Khosravian P., Ghashang M., Ghayoor H. Zinc oxide/natural-zeolite composite nano-powders: Efficient catalyst for the amoxicillin removal from wastewater. Biointerface Res. Appl. Chem. 2016;6:1538–1540.
173. Nair S., Sasidharan A., Rani V.V.D., Menon D., Nair S., Manzoor K., Raina S. Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells. J. Mater. Sci. Mater. Med. 2009;20:S235–S241. doi: 10.1007/s10856-008-3548-5. [PubMed] [CrossRef]
174. Păunica-Panea G., Ficai A., Marin M.M., Marin Ș., Albu M.G., Constantin V.D., Dinu-Pîrvu C., Vuluga Z., Corobea M.C., Ghica M.V. New collagen-dextran-zinc oxide composites for wound dressing. J. Nanomater. 2016;14:7–11. doi: 10.1155/2016/5805034. [CrossRef]
175. Khorasani M.T., Joorabloo A., Moghaddam A., Shamsi H., Mansoori Moghadam Z. Incorporation of ZnO nanoparticles into heparinised polyvinyl alcohol/chitosan hydrogels for wound dressing application. Int. J. Biol. Macromol. 2018;114:1203–1215. doi: 10.1016/j.ijbiomac.2018.04.010. [PubMed] [CrossRef]
176. Rădulescu M., Andronescu E., Cirja A., Holban A.M., Mogoantă L., Bălşeanu T.A., Bogdan C., Neagu T.P., Lascăr I., Florea D.A., et al. Antimicrobial coatings based on zinc oxide and orange oil for improved bioactive wound dressings and other applications. Rom. J. Morphol. Embryol. 2016;57:107–114. [PubMed]
177. Díez-Pascual A.M., Díez-Vicente A.L. Wound Healing Bionanocomposites Based on Castor Oil Polymeric Films Reinforced with Chitosan-Modified ZnO Nanoparticles. Biomacromolecules. 2015;16:2631–2644. doi: 10.1021/acs.biomac.5b00447. [PubMed] [CrossRef]
178. Geraldo D.A., Needham P., Chandia N., Arratia-Perez R., Mora G.C., Villagra N.A. Green synthesis of polysaccharides-based gold and silver nanoparticles and their promissory biological activity. Biointerface Res. Appl. Chem. 2016;6:1263–1271.
179. Khashayar P., Amoabediny G., Larijani B., Hosseini M., Verplancke R., Schaubroeck D., De Keersmaecker M., Adriaens A., Vanfleteren J. Characterization of gold nanoparticle layer deposited on gold electrode by various techniques for improved sensing abilities. Biointerface Res. Appl. Chem. 2016;6:1380–1390.
180. Mikalauskaite A., Karabanovas V., Karpicz R., Rotomskis R., Jagminas A. Green synthesis of red-fluorescent gold nanoclusters: Characterization and application for breast cancer detection. Biointerface Res. Appl. Chem. 2016;6:1702–1709.
181. Nicol J.R., Dixon D., Coulter J.A. Gold nanoparticle surface functionalization: A necessary requirement in the development of novel nanotherapeutics. Nanomedicine. 2015;10:1315–1326. doi: 10.2217/nnm.14.219. [PubMed] [CrossRef]
182. Akturk O., Kismet K., Yasti A.C., Kuru S., Duymus M.E., Kaya F., Caydere M., Hucumenoglu S., Keskin D. Collagen/gold nanoparticle nanocomposites: A potential skin wound healing biomaterial. J. Biomater. Appl. 2016;21:283–301. doi: 10.1177/0885328216644536. [PubMed] [CrossRef]
183. Jayakumar R., Prabaharan M., Sudheesh Kumar P.T., Nair S.V., Tamura H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011;29:322–337. doi: 10.1016/j.biotechadv.2011.01.005. [PubMed] [CrossRef]
184. Regiel-Futyra A., Kus-Liśkiewicz M., Sebastian V., Irusta S., Arruebo M., Stochel G., Kyzioł A. Development of non cytotoxic chitosan-gold nanocomposites as efficient antibacterial materials. ACS Appl. Mater. Interfaces. 2015;7:1087–1099. doi: 10.1021/am508094e. [PMC free article] [PubMed] [CrossRef]
185. Volkova N., Yukhta M., Pavlovich O., Goltsev A. Application of cryopreserved fibroblast culture with au nanoparticles to treat burns. Nanoscale Res. Lett. 2016;11:22. doi: 10.1186/s11671-016-1242-y. [PMC free article] [PubMed] [CrossRef]
186. Martins A.F., Facchi S.P., Monteiro J.P., Nocchi S.R., Silva C.T.P., Nakamura C.V., Girotto E.M., Rubira A.F., Muniz E.C. Preparation and cytotoxicity of N,N,N-trimethyl chitosan/alginate beads containing gold nanoparticles. Int. J. Biol. Macromol. 2015;72:466–471. doi: 10.1016/j.ijbiomac.2014.08.020. [PubMed] [CrossRef] |